Lower dose, less toxic radiopharmaceutical produces better outcomes

Neuroendocrine tumors are cancers that begin in specialized cells called neuroendocrine cells. These cells have traits similar to those of nerve cells and hormone-producing cells. Neuroendocrine tumors, while rare, can occur anywhere in the body. Most affect the cardiothoracic region, eg lungs, appendix, small intestine, pancreas as well as the rectum. There are many types of neuroendocrine tumors: some grow slowly while others develop very rapidly.
Neuroendocrine tumors are characterized by abundant production of somatostatin receptor 2, a naturally circulating hormone that is an important target for scientists studying new treatment approaches.
Peptide Receptor Radionuclide Therapy (PRRT) is the most commonly used treatment for refractive neuroendocrine cancers, delivering cancer-killing radioactive substances directly to tumor sites. This treatment provides symptomatic relief, stopping or slowing tumor growth and improving overall survival for patients. It is a form of molecular therapy that features a protein (called a peptide) which targets cancer cells and which is similar to the naturally circulating hormone, somatostatin. The peptide is mated with a small amount of radioactive material, or radionuclide. They form a radiopharmaceutical called a radiopeptide (177Lu-DOTA-EB-TATE). Injected into a patient, this radiopeptide travels in the bloodstream, finds and then binds to neuroendocrine tumor cells, before delivering a targeted high dose of radiation directly to the cancer cells.
PPRT pharmacokinetics, pharmacodynamics improved
Together with collaborators at the Peking Union Medical College Hospital, the Chinese Academy of Medical Science, Professor Shawn Chen Xiaoyuan of the Department of Diagnostic Radiology, Yong Loo Lin School of Medicine at the National University of Singapore (NUS), has managed to improve the pharmacokinetics (i.e. the absorption and movement of drugs into, through and out of the body) and effectiveness of PPRT, while reducing its dosage and toxicity. The researchers did this by introducing a truncated Evans blue molecule onto octreotate peptide (denoted as 177Lu-DOTA-EB-TATE).
"The EB part allows reversible binding of 177Lu-DOTA-EB-TATEto blood albumin and extends its half-life in the blood, and thus provides an extended therapeutic time window, and improved treatment efficacy over Lutathera," said Prof Chen, who holds concurrent appointments at the NUS Departments of Chemical and Biomolecular Engineering, and the Department of Biomedical Engineering, Faculty of Engineering. Lutathera is a radioactive medication used to treat neuroendocrine cancers.
Clinical trial results
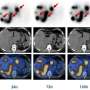
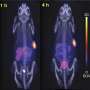
Thirty-two patients at Peking Union Medical College Hospital (PUMCH) with histologically confirmed neuroendocrine tumors were recruited for a clinical trial with the new technology. The patients were randomly divided into three escalating dose groups. The treatments were planned for up to three cycles, repeated at eight-to 12-week intervals. Hematologic parameters, liver function, and kidney function were tested at baseline, one week, and four weeks after each cycle of treatment. The patients were scanned at the start of the trial, some days before the second and third cycles of treatment, and two to three months after their last PPRT cycle.
The patients tolerated 177Lu-DOTA-EB-TATEwell, with good tumor response with almost no side effects. The study was published in the 2021 March issue of the Journal of Nuclear Medicine .
"177Lu-DOTA-EB-TATEwith longer circulation half-life and high tumor accumulation appears to have more potent anti-cancer efficacy than Lutathera, and at a much lower dose. We hope reversible albumin binding through Evans blue derivatives will create various novel therapeutic radiopharmaceuticals that can be effective for not only NET patients but also other types of cancer patients overexpressing different molecular targets," Prof Chen said.
Associate Professor Quek Swee Tian, Head, Department of Diagnostic Radiology at the National University Hospital, said PRRT has been shown to confer a longer progression-free survival and a significantly higher response rate in patients with advanced neuroendocrine cancers as compared to first line medical therapy. "The development of 177Lu-DOTA-EB-TATE, with its improved pharmacokinetics, has further advanced the PRRT therapeutic technology and holds promise for even better therapeutic efficacy and a more efficient workflow for our patients with a drug infusion time a fraction of standard PRRT."
The technology has been licensed to Molecular Targeting Technologies, Inc. (MTTI) and an Investigational New Drug (IND) application has been approved by the U.S. Food and Drug Administration (FDA).
More information: Qingxing Liu et al. Peptide Receptor Radionuclide Therapy of Late-Stage Neuroendocrine Tumor Patients with Multiple Cycles of 177Lu-DOTA-EB-TATE, Journal of Nuclear Medicine (2020). DOI: 10.2967/jnumed.120.248658