Two new over-the-counter at-home COVID-19 tests brought to US market quickly
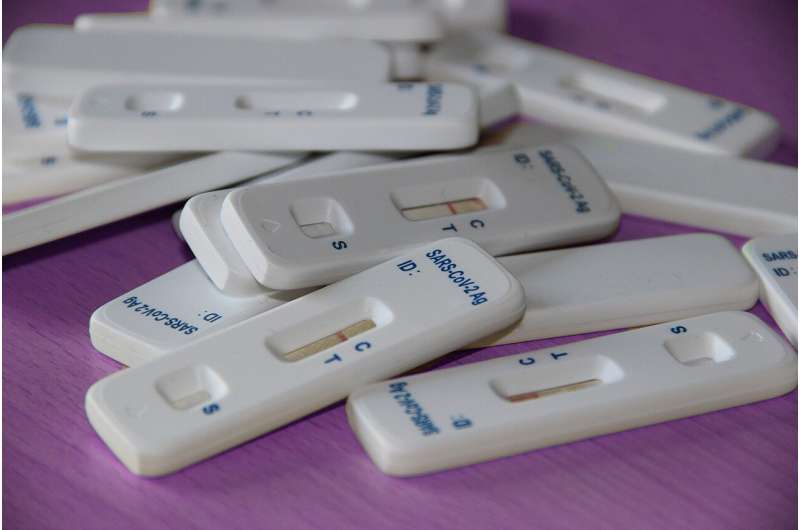
The Biden-Harris administration has brought two new over-the-counter, at-home COVID-19 tests to the U.S. market. The tests, one manufactured by SD Biosensor and distributed by Roche and the other manufactured by Siemens, have received emergency use authorization (EUA) by the Food and Drug administration (FDA) after being evaluated through the administration's new accelerated pathway to support FDA review of tests with potential for large-scale manufacturing that the Department of Health and Human Services (HHS) announced just two months ago.
These quick authorizations are thanks to collaboration between the FDA and the National Institutes of Health (NIH) Rapid Acceleration of Diagnostics Technology (RADx) program. Combined, it is estimated the companies can produce tens of millions of tests per month for use in the U.S.
"Increasing Americans' access to easy-to-use, reliable COVID tests is a top priority for the Biden administration, and we are using all resources at our disposal to make more tests available and ramp up supply," said HHS Secretary Xavier Becerra. "Adding two new authorized tests will give Americans more options for testing at home, which helps keep people safe and provides peace of mind."
In late October, HHS announced the administration would invest $70 million from the American Rescue Plan to help bring more high-quality, at-home tests onto the market in the U.S. in coordination with FDA. This new Independent Test Assessment Program (ITAP) is helping identify manufacturers of high-quality tests and encouraging them to bring those tests to the U.S. market, increasing options for people and overall supply and potentially lowering costs.
In this new program, NIH's RADx Tech network of experts from government, academia, and industry work together with FDA, CDC and other HHS specialists to assess and conduct studies on over-the-counter tests. This coordinated effort allows companies to compile proper data, work towards the right benchmarks for performance, and support other needs that will help ensure they are providing the best submissions possible for FDA's regulatory review. The goal is to accelerate the availability of more high-quality, accurate and reliable over-the-counter tests to the public as quickly as possible. The tests by Roche and Siemens are the first to gain FDA authorization through this program.
"This program is incredibly beneficial to increasing access to rapid tests by quickly and consistently gathering the critical data companies need to request EUA and subsequently enter the U.S. market once authorized," said Jeff Shuren, M.D., J.D., director of the FDA's Center for Devices and Radiological Health. "Knowing these tests have been independently assessed by NIH allows the FDA to act quickly on well-performed studies and trustworthy data that will lead to the availability of additional accurate and reliable tests at this critical time."
"By rapidly conducting the precise studies recommended by the FDA, this program is shaving weeks to months off the typical EUA timeline," said Bruce J. Tromberg, Ph.D., director of the NIH's National Institute of Biomedical Imaging and Bioengineering (NIBIB) and RADx program lead.
This is part of the Biden administration's commitment to increasing access to COVID-19 testing. Last week, President Biden announced new actions to ensure Americans have access to free testing, including convenient, at-home tests. He committed to purchase half a billion at-home tests to be provided to Americans for free this winter, starting in January, and to stand up new federal testing sites across the country. Additionally, there are now 20,000 free testing sites across the U.S., four times as many at-home tests available to Americans than were available this summer, and free at-home tests are already being made available at key community sites, such as community health centers and rural clinics.