MIS-C patients have deeply dysregulated protein profiles, according to new research
Levels of proteins in the plasma of patients with multisystem inflammatory syndrome in children (MIS-C) are significantly dysregulated and share features with two other inflammatory syndromes, according to a new study by researchers at Children's Hospital of Philadelphia (CHOP). The findings were recently published in Nature Communications.
MIS-C emerged as the major pediatric complication of SARS-CoV-2 and is characterized by fever, severe inflammation, cardiac dysfunction, and shock. CHOP researchers previously demonstrated that infection with SARS-CoV-2 and MIS-C are associated with thrombotic microangiopathy (TMA), a syndrome that involves clotting in small blood vessels and has been identified as a potential cause for severe manifestations of COVID-19 in adults. However, the mechanism of TMA in the context of MIS-C is unknown, and its association with cytokine dysregulation remains unclear.
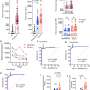
To better understand the cytokine dysregulation in MIS-C and its association with TMA, the researchers analyzed the plasma protein landscape of patients with MIS-C, severe COVID-19, mild or asymptomatic COVID-19, and otherwise healthy controls. Analyzing more than 1,400 plasma proteins, the researchers found that protein signatures of patients with MIS-C overlap with those of patients with macrophage activation syndrome (MAS). In fact, a subset of patients with MIS-C satisfied the modified criteria for the diagnosis of MAS.
Moreover, similarly to MAS, IFN-γ (and one of its downstream proteins, CXCL9) were shown to be dysregulated in MIS-C patients. The researchers also found that levels of IFN-γ may also predict the severity of cardiac dysfunction in patients with MIS-C; paradoxically, they observed that patients who had higher IFN-γ levels displayed better cardiac function than those with lower IFN-γ levels.
Finally, the researchers identified the protein PLA2G2A as a candidate biomarker for MIS-C. They found that levels of PLA2G2A are markedly high in almost all patients with MIS-C and differentiate MIS-C patients from other SARS-CoV-2 infected patients, making PLA2G2A a potential biomarker for MIS-C.
"We have made several observations about the underlying pathophysiology of MIS-C, and our future studies will hopefully identify the mechanistic relationships between IFN-γ, PLA2G2A, and other drivers of MIS-C initiation and immunopathology" said Hamid Bassiri, MD, PhD, an attending physician in the Division of Infectious Diseases at Children's Hospital of Philadelphia and co-senior author of the study, along with David T. Teachey, MD, and Edward M. Behrens, MD. "In particular, it will be important to understand the mechanism of PLA2G2A in MIS-C pathophysiology, since PLA2G2A and its associated pathways can be targeted with the common, inexpensive medication indomethacin."
More information: Caroline Diorio et al, Proteomic profiling of MIS-C patients indicates heterogeneity relating to interferon gamma dysregulation and vascular endothelial dysfunction, Nature Communications (2021). DOI: 10.1038/s41467-021-27544-6