March 1, 2022 feature
The secret's in the spikes: Exploring omicron's ability to escape antibodies
Virologists thought they had seen the worst of SARS-CoV-2's variants when delta emerged in the spring of 2021, but by fall a new one was in circulation with a slew of spike protein mutations that took the scientific community off guard.
"The omicron variant harbors a striking 59 amino acid substitutions throughout its genome relative to the ancestral Wuhan-hu-1 SARS-CoV-2 virus," said Dr. Gene S. Tan, assistant professor at the J. Craig Venter Institute, reporting in Science Translational Medicine.
Working with a team of Venter Institute scientists in La Jolla, California and Rockville, Maryland, Tan and colleagues conducted a series of elegant laboratory experiments to better understand how omicron and other SARS-CoV-2 variants—especially the one known as beta—elude neutralizing antibodies.
They found that the secret to antibody evasion is tucked away in the multiple mutations that pervade each of the viruses. Unlike its cousin variants, omicron is chock-full of mutations, more than any other SARS-CoV-2 variant that has so far caused waves of infection in the pandemic. Among the 59 mutations that characterize omicron, the majority are concentrated in the business end of the virus—the spike, the protein that unlocks human cells to initiate infection.
"Thirty-seven of these mutations are within the spike protein'", said Tan, referring to omicron. And the spikes are the targets of neutralizing antibodies. If the antibodies can't bind to the spikes, they are unable to easily neutralize the virus. "This degree of mutational change raises questions about the effectiveness of neutralizing antibodies that were elicited by infection with SARS-CoV-2 or by current mRNA vaccines," Tan and his colleagues concluded.
The Venter Institute research arrives as scientists worldwide attempt to peel away much of the mystery surrounding SARS-C-oV-2 variants—especially omicron.
Also known as BA.1, omicron is a variant that emerged in the southern region of Africa in late 2021, and it has rocked the world with renewed waves of infection. Even though its surges have already peaked in many parts of the globe, it is still wreaking havoc, forcing healthcare systems to the brink of collapse.
In Hong Kong, so many people have been infected in recent weeks, doctors and nurses are treating patients in parking lots, driveways and sidewalks because they've run out of space in hospitals.
In addition to its multiple mutations, omicron also has sub-variant siblings, such as BA.1.1 and BA.2 that are subjects of new rounds of scientific investigation. As with any infectious virus, omicron has a multitude of tricks: shape-shifting is one, a maneuver that makes it difficult for antibodies to bind to a spike protein's altered conformation. A lavish swathing in glycans (sugar-based polymers) is another. The glycans act as an invisibility coat, obscuring spike proteins from antibodies on the hunt for viruses.
The Venter team, which included lead author Benjamin L. Sievers, collaborated with researchers at Stanford University and the University of Maryland to better understand which SARS-CoV-2 variants were most adept at escaping antibodies. They found that omicron and another variant of concern, beta, were most adept at dodging antibodies.
The findings coincide with discoveries from other researchers globally who've found that escaping antibodies is an evolutionary goal of SARS-CoV-2, and that omicron has proved to be most masterful at antibody escape. Its high degree of contagion, scientists say, has been based largely on its ability to evade the immune system.
To define how efficiently omicron and other variants escape neutralizing antibodies, Tan and colleagues used serum and plasma samples from patients to simulate in the laboratory some of the types of human populations that encounter the virus: people who are vaccinated, those who are vaccinated and boosted, and people who have had a previous SARS-CoV-2 infection. They also took samples from pregnant women because that population is one of the most vulnerable when it comes to SARS-CoV-2 infection. Pregnant women included in the research had previously received two mRNA shots.
Samples for this part of the study were drawn from individuals participating in a longitudinal cohort project and were diagnosed as having had mild SARS-CoV-2 infections. All patients were enrolled in an outpatient study at Stanford Hospital Center. This study was a trial evaluating the efficacy of interferon lambda in mild COVID-19, but only participants from the placebo arm were studied in the Venter Institute investigation.
Omicron wasn't examined in isolation but in comparison to a range of other SARS-CoV-2 variants, including the original Wuhan-hu1-virus, the strain that initiated the worldwide pandemic. Tan and colleagues refer to the Wuhan variant in their paper as D614.
"We evaluated the magnitude and breadth of the antibody response against the D614 virus along with three variants of concern: beta, delta and omicron," Tan wrote. "Understanding these neutralizing antibody responses will enable us to assess the state of pre-existing immunity elicited by the [Wuhan] virus and can inform the design of the next generation of COVID-19 vaccines."
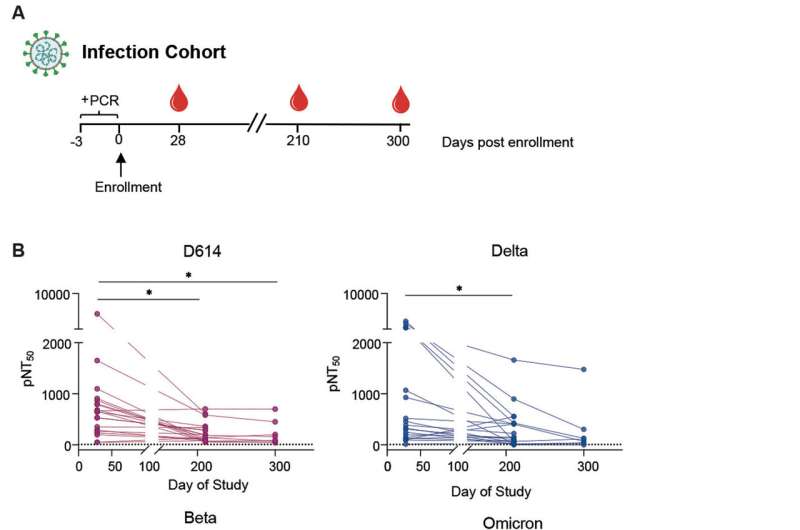
To compare the antibody response—and escape capacity—among SARS-CoV-2 variants, the scientists used a pseudovirus that was coaxed to express the spike proteins and mutations of the SARS-CoV-2 omicron variant. Pseudoviruses were likewise coaxed to express the spike proteins of other SARS-CoV-2 variants, including the original wildtype—or Wuhan-hu1 variant—and the beta and delta variants. These viruses were tested in serum or plasma samples from the groups of individuals to evaluate antibody responses.
"Using vesicular stomatitis virus, VSV, pseudoparticles expressing the spike protein of several SARS-CoV-2 variants, we evaluated the magnitude and breadth of the neutralizing antibody response over time in individuals after infection and in mRNA-vaccinated individuals," Tan wrote in Science Translational Medicine, noting that booster doses increased the magnitude of the antibody response to the Wuhan, beta, delta and omicron variants. However, neutralizing antibody activity remained lowest against omicron, even involving samples from people who were boosted.
"We observed that boosting increases the magnitude of the antibody response to wildtype, beta, delta, and omicron variants. However, the omicron variant was the most resistant to [antibody] neutralization," Tan concluded, based on the research results.
The antibody response was low when beta and omicron variants were tested against samples from people with prior SARS-CoV-2 infections, especially when compared with the delta and Wuhan variants.
The findings arrive as the World Health Organization considered—but declined—giving omicron's sub-variant, BA.2, its own Greek letter. There is no need to parse omicron into different viruses with their own names, WHO's experts in pandemic viruses concluded.
"Omicron is a variant of concern that is dominant worldwide, and we know this from sequencing around the world," said Dr. Maria Van Kerhove, who leads the agency's emerging and zoonotic diseases unit.
"Omicron is less severe than delta, but it is not a mild virus. This is a dangerous virus and there are still some countries that are seeing an overwhelmed hospital system," Van Kerkhove said.
The Venter Institute team, meanwhile, noted findings from their study raised special concerns about pregnant women. There was a lower antibody response in samples obtained during pregnancy, even though the women had received two doses of an mRNA vaccine.
Pregnancy, they underscored, is a risk factor for severe COVID-19. Results from their newly completed research suggest that ways should be sought to increase vaccination among pregnant women, an understudied and vulnerable population.
More information: Benjamin L. Sievers et al, Antibodies elicited by SARS-CoV-2 infection or mRNA vaccines have reduced neutralizing activity against Beta and Omicron pseudoviruses, Science Translational Medicine (2022). DOI: 10.1126/scitranslmed.abn7842
© 2022 Science X Network