Spirulina-based platform for ultra-large-scale production of therapeutic proteins
A common food source has been converted into a biomanufacturing platform that offers a way to rapidly produce mass quantities of biologic drugs for common diseases that currently lack effective treatments. Researchers at Lumen Bioscience report their groundbreaking genetic engineering methods to highly express bioactive proteins in spirulina using large-scale cultivation and processing methods appropriate for biopharmaceutical manufacturing.
The paper also details favorable preclinical and early-stage clinical testing of these manufactured, orally delivered neutralizing antibodies against campylobacter, a major cause of infant mortality in the developing world and a growing antibiotic resistance threat. The product, LMN-101, is now proceeding through Phase 2 clinical trials, and BARDA's CARB-X program is funding development of a second-generation version of this product.
The publication, "Development of Spirulina for the Manufacture and Oral Delivery of Protein Therapeutics," is published in Nature Biotechnology.
"Genetic engineering has transformed the manufacturing of biological products over the past 50 years, yet plant-based biopharmaceutical production has remained an elusive challenge," said Jim Roberts, M.D., Ph.D., lead author and co-founder and Chief Scientific Officer of Lumen Bioscience. "Spirulina-based biomanufacturing combines the safety of food-based biotechnology with genetic engineering methods to produce biopharmaceuticals that may address medical needs that current drug-development tools have failed to solve."
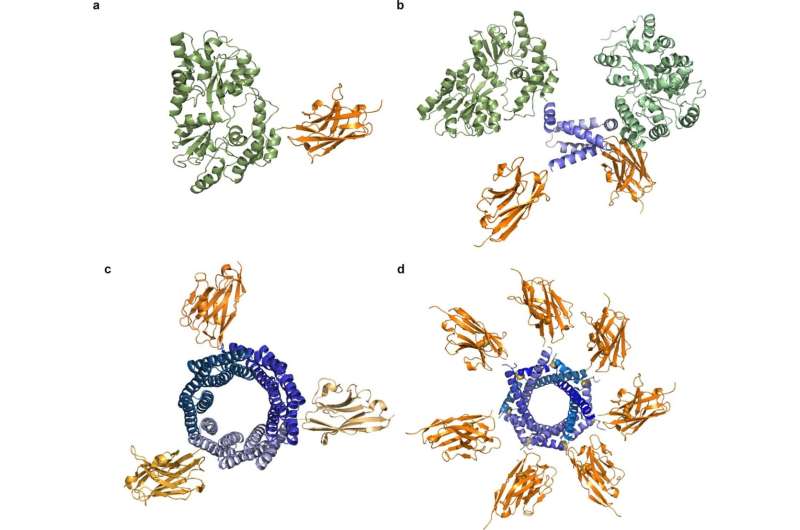
Spirulina, a photosynthetic cyanobacterium, is a highly nutritious food and the only microorganism commercially farmed worldwide as a food. Previous attempts to convert this safe, scalable food source into a biomanufacturing platform have proven futile. In the new paper, Lumen researchers detail methods for the constitutive and stable expression of protein therapeutics in spirulina, with productivities and potencies tens to hundreds of fold higher than achievable in other food-based platforms. These advances in scalability and potency make orally delivered biologics commercially feasible for the first time.
"Delivering therapeutic amounts of protein biologics to the intestine presents an enormous challenge, and for this reason targets in the GI tract have long been considered almost undruggable," Roberts said. "The spirulina platform elegantly addresses this challenge. It can be used to produce and deliver the enormous quantities of ingestible biotherapeutics that will be needed to both prevent and treat gastrointestinal diseases. Preventing and treating diarrheal disease, the example reported in today's paper, offers an excellent case study: these highly prevalent diseases have devastating impacts in the developing world, but traditional drug-making tools have failed to generate a solution despite decades of heavy investment. The approach described in today's publication offers new hope to the individuals exposed to these diseases."
Beyond campylobacter, Lumen is currently applying the technology to a growing list of highly prevalent diseases, including the following publicly disclosed programs:
- LMN-201, a complex biologic cocktail for prevention of C. difficile infection (in collaboration with National Institute of Allergy and Infectious Diseases (NIAID) and Rockefeller University)
- LMN-301, an antibody cocktail for COVID-19 intended for both upper airway and GI delivery (funded in part by the US Army Medical Research and Development Command and the Bill & Melinda Gates Foundation)
- A complex biologic cocktail for treatment of inflammatory bowel disease (targets not yet disclosed)
- In collaboration with Novo Nordisk, orally delivered biologics against GI targets relevant to cardiometabolic disease (targets not yet disclosed)
More information: James Roberts, Development of spirulina for the manufacture and oral delivery of protein therapeutics, Nature Biotechnology (2022). DOI: 10.1038/s41587-022-01249-7. www.nature.com/articles/s41587-022-01249-7