Study shines light on longevity of COVID-19 immune response
By uniting research from eight cohorts across the U.S., a group of researchers has accelerated collection of data integral in answering questions about immune responses needed for long lasting protection from SARS-CoV-2. The description of the cohorts, the assays used and the definitions for events were reported recently in the journal mSphere, an open access journal of the American Society for Microbiology.
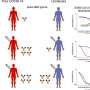
Longitudinal observation studies (studies that employ continuous or repeated measures to follow particular individuals over time) are essential to answer important questions on durability and effectiveness of immune responses against SARS-CoV-2. Often individual cohorts have limited longitudinal data or participant numbers to draw robust conclusions. To overcome these limitations, harmonized yet independent cohorts were established in different geographic locations of the U.S.—the PARIS (Protection Associated with Rapid Immunity to SARS-CoV-2) and SPARTA (SARS SeroPrevalence and Respiratory Tract Assessment) cohorts. These cohorts offer a nimble and rapid approach to stand up a network suitable to collect biospecimens and other data.
"We started the PARIS cohort at the Icahn School of Medicine at Mount Sinai because we wanted to know three things: how long antibody responses to SARS-CoV-2 last during infection, whether antibody responses protect against reinfection and how much antibody an individual needs to be protected," said senior study author Florian Krammer, Ph.D., a professor of Microbiology and Pathology at the Icahn School of Medicine at Mount Sinai.
"It is very important when you have an emerging virus to study the immune responses to the infection itself over time, to look at the protective effects of immune responses induced by infection, and also study what happens when people get vaccinated," added lead study author Viviana Simon, M.D., Ph.D., a professor of Microbiology, Infectious Diseases and Pathology at the Icahn School of Medicine at Mount Sinai.
In the new study, to understand re-infection rates and correlates of protection for SARS-CoV-2, the researchers established eight different longitudinal cohorts in 2020 under the umbrella of the PARIS /SPARTA studies. The cohorts included seropositive and seronegative participants at high risk for infection. The initial PARIS study focused on health care workers in New York City, one of the early epicenters of the pandemic in the U.S., but the other cohorts targeted not only healthcare workers, but other populations including communities of color, first responders and students.
With the rapid SARS-CoV-2 vaccine rollouts starting in mid-December 2020 in the United States, many of the cohorts also now track immune responses to vaccination in individuals both seronegative and seropositive at the time of immunization. There were 8,741 participants in the eight cohorts. In all eight cohorts, the researchers followed individuals with and without COVID-19 by collecting data as well as biospecimens to measure immune responses (e.g., antibody responses to the spike protein of SARS-CoV-2) at least every two months.
"We saw that the antibody response in previously infected individual was relatively stable, and they were protected from re-infection unless the new infection was the Omicron variant," said Dr. Krammer. The team studied how immune responses behaved in previously infected individuals versus those who hadn't yet been infected. The researchers showed that previously infected individuals mounted very rapid immune responses even after a single vaccine dose. "Vaccination boosts your protection and provides better immunity," said Dr. Krammer.
More information: Viviana Simon et al, PARIS and SPARTA: Finding the Achilles' Heel of SARS-CoV-2, mSphere (2022). DOI: 10.1128/msphere.00179-22