First patient receives Moderna mRNA trial infusion for glycogen storage disease
Prior to COVID-19 vaccines most people had never heard of messenger RNA (mRNA) used to create two of the COVID-19 vaccines on the market. However, mRNA was discovered in 1960 and since that time has been researched and used for a variety of vaccines and treatments.
Moderna has been researching the use of mRNA for the potential treatment of the metabolic disorder Glycogen Storage Disease Type Ia (GSD-Ia) caused by an enzyme deficiency where the liver fails to break down glycogen into glucose, causing the body's blood sugar levels to drop. The disorder is devastating, causing life-threatening low blood sugar, and potential damage to kidneys and liver along with other serious side effects, including death.
When Loveah Hernandez, of Dallas, Texas, was six months old she suddenly became unresponsive and was taken to the Emergency Department at a nearby hospital. A week later she had a liver biopsy and was diagnosed with GSD-Ia.
Hernandez had a feeding tube inserted in her nose so she could receive the vital nutrients she needed from the cornstarch therapy, which is used as a slow-release form of glucose necessary to keep GSD-Ia patients alive. Then, when she turned 3 years old, the tube was removed and she received a gastrostomy tube, often called a G-tube, surgically placed to give direct access to her stomach for supplemental feeding.
Like other GSD-Ia patients, Hernandez needs regular cornstarch therapy every 3-4 hours to stay alive. This means sleepless nights and her mother's daily visits to the school to make sure she stayed on the schedule. As she grew older, she started drinking the corn starch mixture mixed with a sugar-free fruit punch flavored mix.
Her life has been significantly impacted by this rare disease with the loss of school trips, lack of invitations to birthday parties because the children were afraid of the g-tube, and regular visits to the hospital when she was sick.
"A large part of my childhood was spent in the hospital," says Hernandez. Although a highlight of that was when The Band Perry came to visit and Kimberly Perry gave her a guitar pick she still has today.
Hernandez, now 21, graduated fourth in her high school class and is now a biology major in college with plans to continue on to become a physician's assistant.
She has been trying for two years to be a trial participant and worked hard to make sure she met the criteria to participate in the Ba1ance Moderna Trial.
The Ba1ance Trial aims to see if a new investigational medication, mRNA-3745, for people with GSD-Ia is safe and if it could potentially help correct the defect that causes GSD-Ia and restore function. If successful, mRNA-3745 would teach certain cells in the body to effectively break down glycogen, correct low blood glucose, and avoid starch intake.
The mRNA technology uses messenger ribonucleic acid (mRNA), an instructional molecule that naturally occurs in the body and carries information to cells. In this case, Moderna is researching whether using mRNA can instruct the body to make the protein that is missing in people with GSD-Ia.
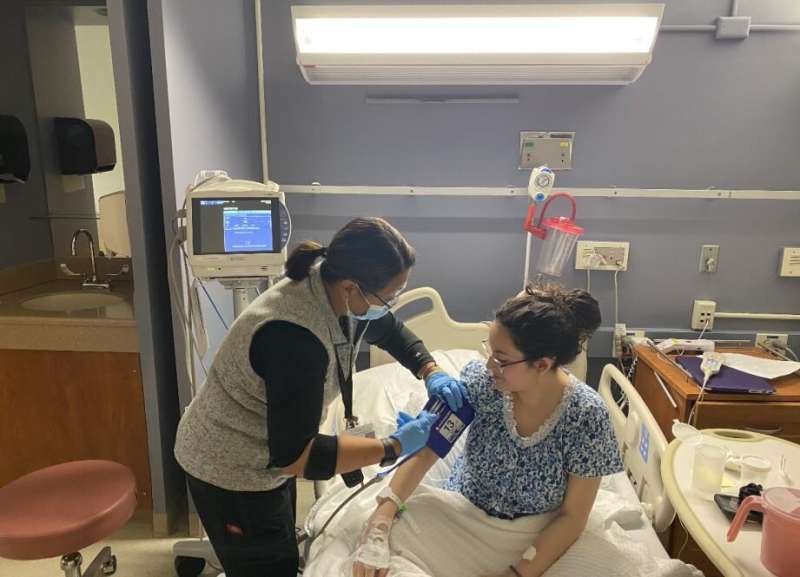
In this phase 1 clinical trial, mRNA-3745 is administered as a single intravenous infusion via a small tube inserted into a vein in the arm.
After a year of planning and preparing, on June 1, 2022, Hernandez was the first person in the world to receive the investigational mRNA-3745 infusion.
"It's pretty cool to be the first person to receive this investigational treatment," says Hernandez.
"Finding the right patient to participate is difficult because many don't want to be the first," says Shaylee King, clinical research manager, Department of Pediatrics at UConn Health. "I am in awe of Loveah who has shown her selflessness and bravery to be the first to participate in this trial in an effort to potentially provide another treatment option for those with GSD-Ia."
"This trial is exciting and historic. We know mRNA vaccines work. They have been critical to the fight against COVID-19. Now we can potentially use the same technology to help manage a completely different kind of illness," says Juan C. Salazar, MD, physician-in-chief, Connecticut Children's and department chair of Pediatrics at UConn School of Medicine. "This could be a life-changing treatment for so many children and their families across the globe. It's truly spectacular."
The conversations with Moderna and this trial started over four years ago and in mid-July 2021 they we ready to start the trial with UConn Health and Connecticut Children's.
"It's a testament to our team that their experience with first in human GSD-Ia trials provided the skills and processes to move this trial forward quickly and efficiently," says Dr. Bruce Liang, interim CEO of UConn Health and dean of the UConn School of Medicine. "The fact that we are doing it here shows how far UConn Health, the Department of Pediatrics at the UConn School of Medicine, in partnership with Connecticut Children's, has come as a research enterprise."
Karen J. Loechner MD, Ph.D., pediatric endocrinologist at Connecticut Children's, is the Principal Investigator (PI) for the Ba1ance Trial with 20 years of research experience. The Co-PI of this study is Dr. Narinder Maheshwari from the Department of Medicine at UConn Health.
Loechner explained that the purpose of Phase 1 of the Ba1ance Trial is to determine if the investigational treatment, mRNA-3745 is tolerable, safe and what effects it has on the blood sugar during fasting. It is hoped that the investigational mRNA treatment will provide the missing code to permit the liver to make the defective protein of GSD-Ia.
"The goal is to provide an improved quality of life for GSD-Ia patients so they are not dependent on corn starch therapy," says Loechner.
Most rare diseases don't gain a lot of attention and research so this study gives Hernandez hope for herself and those with GSD-Ia.
"My hope for this trial is that it potentially provides a successful treatment to improve my life and the lives of others with GSD-Ia," says Hernandez.
The Ba1ance Trial includes one overnight stay at a hospital and four additional visits at the hospital or trial doctor's office. If agreed to by the participant and trial doctor, all other visits will be at the participant's home where a home health nurse will perform the assessments needed.