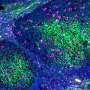
Cancer cells adopt hitherto unknown state to facilitate metastasis
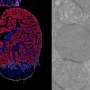
The ancient Egyptians, as described in the Ebers Papyrus, already knew that palpation—feeling for hardened lumps—can help diagnose breast cancer. Palpation is still an important element in early screening for breast cancer. On the other hand, measurements on individual cancer cells show that they are softer than the healthy epithelial cells from which they stem, which probably makes them better able to metastasize in dense human tissue. An international collaborative project led by the Soft Matter Physics Division at Leipzig University got to the bottom of this apparent paradox and has now published its findings in the journal Nature Physics.
In order to grow in healthy tissue, a tumor must displace the surrounding tissue. To do this, it must generate mechanically stable resistance in order to gain space. On the other hand, a high degree of deformability of individual cancer cells is advantageous for invading other tissue. These conflicting demands are met in malignant tumors as the cancer cells become mechanically more heterogeneous and softer, adopting a previously unknown new material state that combines the advantages of solids and liquids.
Physicist Dr. Thomas Fuhs from the Soft Matter Physics Division at Leipzig University, who was lead author on the study, explains, "Breast and cervical tumor patient samples show a greater spectrum of mechanical properties, which pushes the cancer cells toward softer cells, even though the entire tumor remains a solid mass."
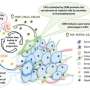
Motivated by computer simulations, in cancer cell clusters the researchers found small islands of rigid, immobile cells surrounded by a multitude of motile, soft cells. The soft cells can move freely, almost like a liquid, and the stiff cells block each other's movement, jamming each other like coffee beans in a funnel. The cancer cell clusters in a tumor form a new material state in which only a few solid islands of hard cells in a fluid environment of soft cells are sufficient for the tissue to behave as a mechanically stable solid rather than as a liquid, despite a majority of mobile, soft cancer cells. The islands of hard cells are connected by mechanical stress bridges of soft cells. This state stabilizes the tissue sufficiently to allow tumor growth, while providing plenty of room for soft, motile cells to break out of the tumor to form metastases.
"The paradox that, in breast tumors, cells that become softer actually form a structure that is harder than the original tissue is only an apparent contradiction. This effect is further enhanced because here mainly very soft fat cells in the healthy breast are compared with cells that are softer than healthy epithelial cells, but still significantly harder than fat cells," explains Professor Josef Alfons Käs from Leipzig University.
In other carcinomas, such as cervix carcinomas, there is no difference in the macroscopic stiffness of healthy and tumor tissue. Yet even in these carcinomas, there is a broader distribution of individual cancer cell elasticity, leading to the new mechanical state that allows a mechanically stable tumor to contain a high degree of motile cells capable of metastasis. The researchers assume that this is probably true for all solid tumors, and thus 92% of all cancer patients.
"The results of our study provide essential new insights into tumor biology or more precisely into the metastatic cascade. The new insights into the mechanics of cancer cells and tumor tissue demonstrate that the physics of cancer is an important element in understanding tumor progression," explains Fuhs, who adds that whether the cells in a tumor remain completely jammed, as in healthy tissue, or are able to unjam due to the softening of the cancer cells can make all the difference for whether a tumor metastasizes.
More information: Thomas Fuhs et al, Rigid tumours contain soft cancer cells, Nature Physics (2022). DOI: 10.1038/s41567-022-01755-0