This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Researchers achieve gene therapy milestone for potential Cockayne syndrome treatment
Researchers working with UMass Chan Medical School's Translational Institute for Molecular Therapeutics announced progress in developing a vector to deliver gene replacement therapy in mice models with Cockayne syndrome, a rare and fatal neurodegenerative disease that largely affects children and young adults.
The proof-of-concept milestone for the adeno-associated virus (AAV) vector gives hope to parents such as Jo Kaur and Richard DiGeorge of New York, who are desperately seeking a cure for their child. The couple founded a nonprofit organization, Riaan Research Initiative, and entered into an agreement with UMass Chan in 2021 to support research after their son Riaan was diagnosed with the fatal autosomal recessive disorder.
Ana Rita Batista, Ph.D., instructor in neurology, is leading the research, along with Miguel Sena-Esteves, Ph.D., associate professor of neurology and director of the Translational Institute for Molecular Therapeutics.
"It is very, very exciting," said Kaur. "This development gives us a lot of promise and hope that we can translate that treatment that we see working in mice into children like Riaan and others around the world who are suffering and really don't have a lot of options. Thanks to the highly motivated and brilliant UMass Chan team, we have reached a groundbreaking milestone for the Cockayne syndrome community, in a very short time."
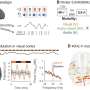
"We now have AAV vectors that actually have a profound impact in the survival of the animals and right now they appear to be normal, the ones that have been treated," said Dr. Sena-Esteves. "So, the progress has been very good. And obviously, our goal ultimately is to move toward clinical trials."
Dr. Batista explained that Cockayne syndrome is caused by genetic mutations in either the ERCC8 (CSA) or ERCC6 (CSB) genes. The most common characteristic of the disease is the very small size of the child. Many will have developmental delays and vision and hearing problems, among other things, but there is a broad spectrum of impacts.
"What we're working on now is developing a gene therapy approach where we'll be giving these patients a normal functional CSA gene that hopefully will improve their life," Batista said.
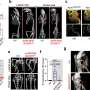
The research team reported that in initial studies they have extended the lifespan of the animal models, which resumed normal growth after treatment, according to Sena-Esteves.
He continued, therapies for ultrarare diseases such as Cockayne—which afflicts fewer than 5,000 individuals in the United States according to the National Institutes of Health—are hard to make a commercial case for because of the high cost of research and development with relatively low anticipated market revenue. That's why partnerships with family fundraising organizations such as the Riaan Research Initiative are so important.
The Translational Institute was launched in 2022 to streamline the process and lower the upfront costs associated with developing gene therapies for rare diseases.
"This is sort of a pipeline that we're building and putting in place so that we can move these therapies faster to the clinic," said Sena-Esteves. "The proof-of-concept experiments in mice are necessary to prove to us that it's worthwhile moving forward."
Meanwhile, 3-year-old Riaan provides a personal face to the scientists, as they search for effective therapies. He is bright, energetic, loves playing soccer using his gait trainer and listening to Sesame Street and Cocomelon songs, according to his parents. "Most people think, oh, he has a rare disease, he won't be able to do certain things, but it's actually the opposite," DiGeorge said.
Sena-Esteves said that while one goal is to create capacity through the institute, "Mostly, the goal is to serve the rare disease community, so we can have an impact and change a little bit the paradigm in which this operates, this translation from the bench to the clinic. Unless something changes, these diseases are going to be left behind."