This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Short dose of intravenous sedative may prevent postoperative delirium after heart surgery
Postoperative delirium—a behavioral state characterized by confused thinking and a lack of awareness—occurs in 10–30% of older patients who are recovering from heart surgery, and it has been linked with long-term cognitive deficits, prolonged hospitalization and institutionalization, and an increased risk of early death, resulting in total attributable health care costs of approximately $32.9 billion annually.
Results from a recent clinical trial led by scientists at Massachusetts General Hospital (MGH) indicate that nighttime treatment with the sedative dexmedetomidine following surgery may help to prevent postoperative delirium in these patients.
The trial, which is published in eClinicalMedicine, included patients older than 60 years of age who were scheduled to undergo a cardiac surgical procedure at MGH with planned postoperative admission to the cardiac surgical intensive care unit (ICU).
A total of 394 patients were randomized in the Minimizing ICU Neurological Dysfunction with Dexmedetomidine induced Sleep (MINDDS) trial to receive a short nighttime dose of intravenous dexmedetomidine (1 μg/kg over 40 minutes) or placebo.
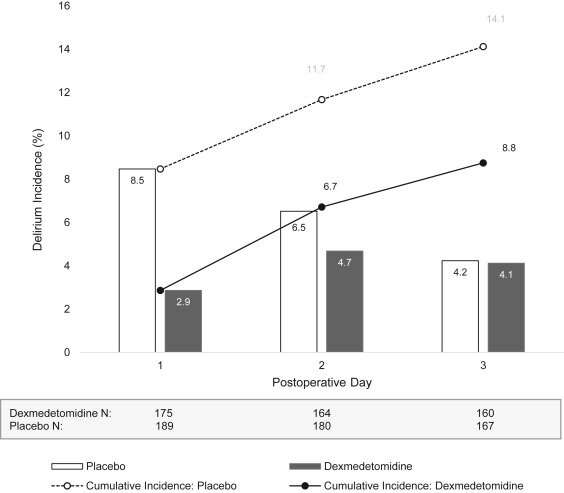
Among patients in whom postoperative delirium status could be assessed, delirium on postoperative day one occurred in 5 of 175 patients (2.9%) in the dexmedetomidine group and 16 of 189 patients (8.5%) in the placebo group, representing a 68% lower odds of delirium in the dexmedetomidine group.
Also, when the cumulative incidence of delirium was studied, a non-significant but higher proportion of participants experienced delirium within three days postoperatively in the placebo group (14.1%) compared with the dexmedetomidine group (8.8%). Long-term cognitive function, quality of life measures, and mortality rates were similar between the groups.
"At present, definitive guidelines or large clinical trials do not support the use of any medication for postoperative delirium prevention," says senior author Seun Johnson-Akeju, MD, Anesthetist-in-Chief and the Henry Isaiah Dorr Associate Professor of Research and Teaching in Anesthetics and Anesthesia at MGH. "However, our data support nighttime dexmedetomidine as a prophylactic treatment to reduce the public health burden of postoperative delirium and as a possible strategy to improve the ICU patient experience."
When describing their future plans, co-author Ariel Mueller, MA, Administrative Director for Research at MGH, stated that "thanks in large part to the infrastructure established by Dr. Johnson-Akeju and lead author Dr. Jason Qu between the anesthesia and cardiac surgery departments, this innovative study not only answered an important question, but it also set the stage for future research in this arena. With support from MGH's Anesthesia Research Center, we are currently planning a multicenter study that we hope to start next fall."
More information: Jason Z. Qu et al, Nighttime dexmedetomidine for delirium prevention in non-mechanically ventilated patients after cardiac surgery (MINDDS): A single-centre, parallel-arm, randomised, placebo-controlled superiority trial, eClinicalMedicine (2022). DOI: 10.1016/j.eclinm.2022.101796