This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Examining the factors influencing participation of Black and White Americans in Alzheimer's disease biomarker research

The use of Alzheimer's disease (AD) biomarkers such as amyloid and tau levels to facilitate early detection and improvement of treatment of AD is a high-priority research area. However, AD biomarker research necessitates diverse research participants given that the levels of biomarkers and their clinical interpretations may vary based on race. Yet, Black Americans are underrepresented in AD research despite having higher risks of developing AD compared to White Americans.
A new study, led by U.S. Department of Veterans Affairs and Regenstrief Institute Research Scientist Johanne Eliacin, Ph.D., examines barriers to participation and to drivers of engagement in AD biomarker research. The study involved interviews with Black and White older adults in the Indianapolis metro area who are new to research. It explored their views of barriers to AD biomarker research participation and factors that may motivate them to engage in research.
Participants in the study discussed how racial bias and healthcare disparities undermine their trust in research. This theme was especially prominent in interviews with Black individuals.
"Although previous work has shown that individuals of African American descent are as willing as White individuals to be involved in research, it's been a persistent problem that Black individuals are underrepresented in clinical trials, especially clinical trials that focus on Alzheimer's disease," said Dr. Eliacin.
"When people don't know what the study involves, the purpose of the research, or why their participation is worthwhile, it's hard for them to volunteer their time. We need to do a much better job of communicating the importance of research participation."
"The under-representation of Black Americans in Alzheimer's research threatens to exacerbate existing health disparities. We need to demonstrate the need for Alzheimer's biomarkers research and the compatibility of research participation with their values and culture."
In the study, a participant commented on how lack of trust is a barrier to research participation and the value of relationship in research engagement. In this paraphrase, one participant explains: There's no way that I think anyone would have surgery if you just talked to a nurse, to a secretary…and didn't meet that doctor. I don't know why in research situations the research doctor doesn't think a person wants to talk to him or her… If I can't connect with you in conversation, I can't allow you to experiment on my body.
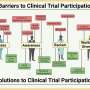
Overall, participants in the study identified four key barriers to AD biomarker research participation:
- hesitancy due to fear, including fear of the unknown about research and fear of potential AD diagnosis.
- hesitancy due to distrust of research and researchers.
- lack of knowledge about AD and related clinical procedures.
- lack of disclosure of research test results.
Drivers identified by study participants for research engagement:
- knowledge of research, AD and related clinical procedures.
- perceived benefits to individuals and community.
- outreach from trusted sources.
The number of individuals in the U.S. diagnosed with AD is expected to increase to 12.7 million by 2050.
"This work is so important, because increasing research participation has huge implications," said study co-author Sophia Wang, M.D., an assistant professor of psychiatry at Indiana University School of Medicine, who sees patients living with Alzheimer's and notes the importance of early AD diagnosis.
"We'll be modifying strategies to engage Black individuals in response to what we've learned from our work. This includes developing materials that they want to see and getting information from who they trust most and in a format that makes sense to them. The field is changing at an unbelievable pace, literally on a daily or weekly basis. So, working closely with communities, earning their trust to make sure they get the most reliable information so they can make good decisions about themselves and their loved ones who are affected by this disease, is critically important."
Participants in the study were one multi-race, 10 White and 15 Black individuals, all age 55 or older, a majority of whom had at least a four-year college education. Many participants, from diverse racial backgrounds, who expressed interest in participating in AD biomarker research, reported prior positive research experiences, familiarity with clinical procedures, and/or a healthcare background.
The authors conclude, "In summary, as with medical decision-making, personal decisions about research participation often hinge on a perceived risk-benefit analysis. Our fundings illustrate how one's individual experience may influence the identified risks and benefits of AD biomarker participation. Culturally attuned engagement strategies may amplify those perceived benefits and diminish the perceived risks by building knowledge and bridging the barriers that separate the research community from potential participants."
The research is published in Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring.
More information: Johanne Eliacin et al, Factors influencing the participation of Black and White Americans in Alzheimer's disease biomarker research, Alzheimer's & Dementia: Diagnosis, Assessment & Disease Monitoring (2022). DOI: 10.1002/dad2.12384