This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers show how bending implantable medical devices can lead to bacterial growth
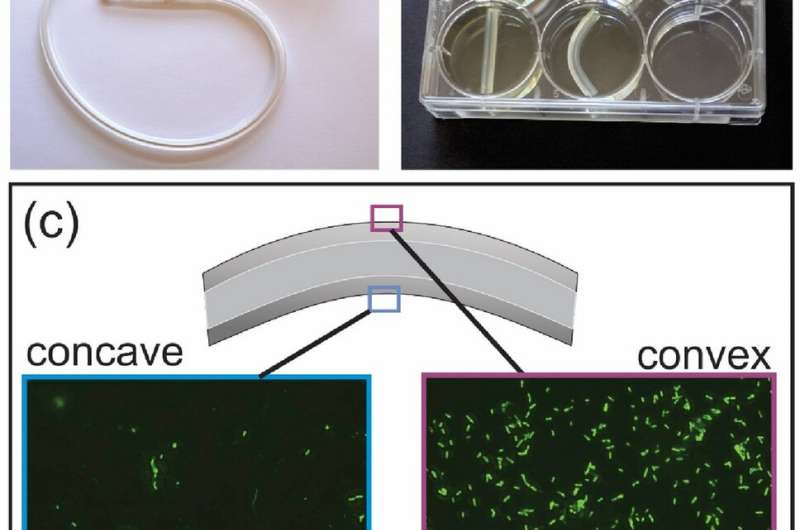
A study by researchers at the University of Toronto's Faculty of Applied Science and Engineering shows that mechanical deformation of medically implantable materials—such as bending or twisting—can have a big impact on the formation of potentially harmful biofilms.
The study, described in a paper published in Scientific Reports, shows that even slight bending of elastomeric materials such as polydimethylsiloxane (PDMS)—also known as silicone—opens up microscopic cracks that are perfect environments for colonizing bacteria.
"These kinds of materials are used in all kinds of biomedical applications, from catheters to tracheal tubes and prosthetic breast implants," says Ben Hatton, associate professor in the faculty's department of materials science and engineering, and senior author of the new study.
"The formation of microbial biofilms on these materials is common, but we were surprised by the degree to which bending silicone, and other rubber materials, causes these cracks to reversibly open and close—and how big a difference they make in terms of biofilm formation."
Biofilms are complex communities of organisms that grow on surfaces. While individual microbial cells are susceptible both to antibiotics and the body's natural defensive systems, the biofilm environment can shield them from these interventions, which can lead to persistent infections.
Infections associated with medical-device biofilms, which sometimes develop after surgery, can be serious health risks—lengthening hospital stays or causing patients who have been discharged to be readmitted.
Hatton and his team are among many groups around the world developing new materials, coatings and other approaches to prevent the formation of biofilms that can lead to such infections.
But in their latest work, they chose to study something more fundamental: How do these colonizing microbial organisms gain a toehold in the first place?
"In part, this comes out of the multidisciplinary approach we take in our group," Hatton says.
"We're combining microbiology and materials science, but also mechanical engineering, because we're talking about mechanical stress, strain and deformation. This bending effect is something that had not been noticed before."
The team tested various samples of silicone, including some they synthesized themselves as well as commercial-grade medical tubing used for urinary catheters. They then subjected these samples to mechanical forces to create surface damage. Their experiments showed that the microcracks can be formed very easily.

"One thing we did was simply wipe them a bunch of times with an ordinary laboratory tissue," says Desmond van den Berg, a Ph.D. candidate in the Institute of Biomedical Engineering and lead author on the paper, which was co-authored by fellow Hatton Lab researchers Dalal Asker and Tarek Awad.
"Even this wiping was enough to create surface damage. By eye it still looks fine, but under the microscope, we could already see microcracks of the size that bacteria could get into. Bacteria are only a few micrometers big, so it doesn't take much."
Other samples were pressed with a rough, ridged pattern to create a series of regularly-spaced microcracks.
All the samples were then placed in a bacterial culture plate and seeded with Pseudomonas aeruginosa, a biofilm-forming bacterium commonly used as a model organism in these types of studies. After growth, the samples were treated with a fluorescent dye, causing any attached bacteria to glow green under an optical microscope.
"What we saw was that the bacteria very clearly preferred to attach in these microscopic cracks," van den Berg says.
"In the bent samples, there were four to five times as many bacteria on the side that was in tension versus the side that was in compression. These cells have full choice about where to grow, but they clearly love the side where all these microcracks are opened up."
The only samples that remained relatively free of bacteria were the ones that had been synthesized in the team's own lab, and therefore remained extremely smooth, van den Berg notes.
"Even the commercially produced ones that we didn't damage in any way already had microcracks in them, straight out of the package," he says.
"We suspect that these are introduced via the manufacturing process, arising from the way the plastic is formed into tubes or other shapes by extrusion or injection molding."
Hatton says that the study underlines just how easy it is to introduce surface features that encourage microbial attachment. For surgeons, one implication is that they should be looking out for situations where silicone devices—such as tubing or prosthetic implants—are being bent during use, and paying particular attention to the side under tension, as this is where infections might be starting.
"Obviously it's difficult to simply not bend a rubber tube that is meant to be bent—otherwise, why would you make it out of rubber in the first place?" Hatton says.
"But perhaps we can learn more about how to control or hide these surface cracks, so that bending is not a problem. That's what we're working on now—researching methods to reduce surface damage, or modifying the silicone surface to reduce the formation of such cracks."
More information: Desmond van den Berg et al, Mechanical deformation of elastomer medical devices can enable microbial surface colonization, Scientific Reports (2023). DOI: 10.1038/s41598-023-34217-5