May 30, 2023 report
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Nitric oxide levels found to have reversible causative role in autism spectrum disorder in a mouse model
Researchers at the Hebrew University of Jerusalem and the University of Haifa in Israel have discovered that production of nitric oxide in the brain correlates with autistic symptoms. In their paper, "The NO Answer for Autism Spectrum Disorder," published in Advanced Science, the researchers describe how an initial observation in a mouse study led to a deeper investigation into the activity of nitric oxide and the discovery of a quantity-dependent causative correlation to disease pathology.
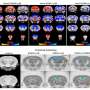
The study results show that nitric oxide (NO) has a role in autism spectrum disorder (ASD) development, affects synaptogenesis and the glutamatergic and GABAergic systems in the cortex and the striatum, converging into ASD-like behavioral deficits. The experiments establish a direct link between NO and ASD, confirmed in animal models, human iPSC-derived cortical neurons, and clinical samples.
Autism spectrum disorder is a neurodevelopmental and behavioral disorder that typically manifests in childhood. It is characterized by abnormalities in social interactions, deficits in communication, restricted interests, and repetitive behavior, affecting 1 in 44 children globally.
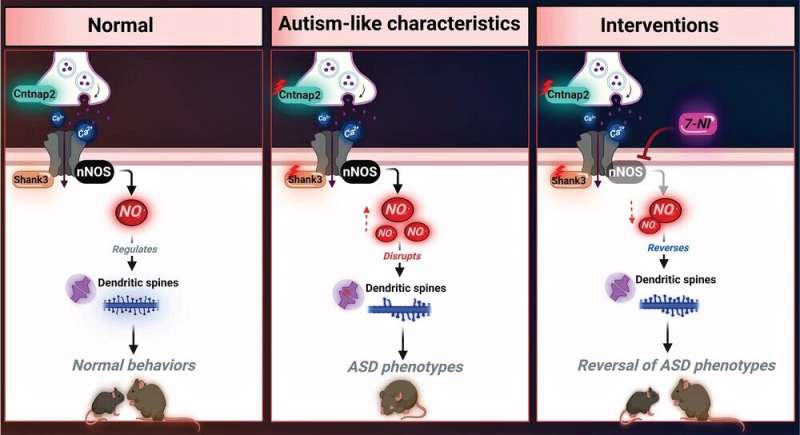
Specific gene pathologies have been correlated with ASD. Deletions or mutations in the SHANK3 gene and CNTNAP2 genes with loss-of-function mutations are highly implicated. When researchers find genetic correlations to a disease, it suggests that a mechanism related to how those genes are meant to function normally has been disrupted. The molecular basis for those disruptions can be more challenging to identify.
In past research with mice genetically altered to mirror ASD, the research team detected elevated nitric oxide levels. NO is a multifunctional signaling molecule and neurotransmitter that regulates cell survival, differentiation and proliferation of neurons as well as synaptic activity, plasticity and vesicle trafficking.
To determine whether NO can lead to ASD-like phenotypes, mice were treated with S-nitroso-N-acetyl penicillamine for ten consecutive days. This increased NO production, resulting in a significant reduction in the cortical dendritic spine density in the mice, similar to what is seen in the genetically altered ASD mice.
Next, the researchers tried the experiment the other way around, inhibiting NO formation in mice modified to mirror ASD conditions. In this group, treatment restored the dendritic spine numbers, potentially reversing some genetically-driven ASD effects.
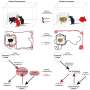
While encouraging, these biochemical results might not necessarily translate into behavioral phenotype changes in the mice. So the researchers recreated the experimental conditions and put modified mice through behavioral tests.
A novel object recognition (NOR) test showed that the normal mice spent significantly more time exploring the novel object than the familiar object. In contrast, the ASD-modified mice did not prefer novel objects over familiar ones. This suggests that the ASD group had deficits in novelty-seeking and memory. Following treatment with a NO inhibitor, ASD modified mice significantly preferred the novel object over the familiar object.
The study findings of a mechanistic link between nitric oxide levels and autism spectrum disorder are significant. It is the sort of substantial discovery typically followed by a caveat-filled word salad suggesting that it might lead to a pathway for future researchers to eventually stumble upon a key to unlock the door of potential clinical treatments.
In this case, by showing a reversal of pathology through specifically targeting a single mechanism while testing a hypothesis, the researchers may have taken the search for a clinical treatment target right to that door with key in hand.
More information: Manish Kumar Tripathi et al, The NO Answer for Autism Spectrum Disorder, Advanced Science (2023). DOI: 10.1002/advs.202205783
© 2023 Science X Network