This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study completes most in-depth mapping of healthy gut bacteria transfer from mother to infant
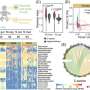
![Analysis of both the microbiota and cultured isolates in the study. a Overview of samples analyzed by shotgun metagenomics (top white) and bifidobacterial culture isolations (bottom gray). Maternal samples are represented to the left of “|” while infant samples are on the right. The timepoint of sampling is shown across the top [Created with BioRender.com]. b The prevalence and relative abundance of the total Bifidobacterium genus in maternal (n = 129, green) and infant (n = 119, gray) stool as determined by metagenomic sequencing is represented in the left plots. Stratification for the 4 most dominant Bifidobacterium species is adjacent. c Clustering of the microbiome of all sequenced samples (n = 1011) based on Bray-Curtis is shown by the dendrogram. Each block represents a different sampling timepoint as shown in the first colored annotation bar. Further relevant covariates are annotated in subsequent color bars. The alpha (Shannon) diversity of each sample is represented in the green dot plot. d Total number of culture-isolated and sequenced (n = 489) Bifidobacterium species isolated from breast milk, maternal, and infant stool. e Phylogenetic tree of all Bifidobacterium strains isolated in this study together with type strains for all bifidobacterial species. Each colored clade highlights the different species isolated. Credit: Nature Communications (2023). DOI: 10.1038/s41467-023-38694-0 Study completes most in-depth mapping of healthy gut bacteria transfer from mother to infant](https://scx1.b-cdn.net/csz/news/800a/2023/study-completes-most-i.jpg)
Researchers have completed the most in-depth investigation to date of mother-to-infant transmission of common gut bacteria. The study reveals the effective transfer of the gut bacteria Bifidobacterium, a major component of health from infancy through to adulthood.
The MicrobeMom joint research project is led by researchers at APC Microbiome Ireland, a world-leading SFI research center based in University College Cork (UCC) and Teagasc, PrecisionBiotics Group, the UCD Perinatal Research Center, University College Dublin, and the National Institute for Bioprocessing, Research & Training (NIBRT).
The MicrobeMom project, shows that maternal-to-infant transmission of gut bacteria is a common phenomenon and is strongly influenced by external factors including mode of delivery and exposure to antibiotics in labor. The study is published in Nature Communications.
Researchers found that Bifdobacterium—a probiotic or healthy bacteria—are present from early stages in infant gut with a range of health and functional benefits, including reducing the development of allergies and asthma. The findings could support the development of targeted probiotic supplements based on these bacteria, with potential benefits to boost immune systems, increase microbial infection resistance, fight disease and aid digestion.
The study detected that delivery mode is a key factor in effective strain transmission with greater microbe diversity observed in vaginal births, with the highest transfer rates linked to spontaneous labor. Maternal exposure to antibiotics during labor significantly reduced the number of strains shared.
MicrobeMom commenced in 2017 to investigate the most likely methods of transfer of bifidobacteria strains from mother to baby, and impact of the mother's diet and health on her gut bacteria.
An earlier study, published in the American Journal Of Obstetrics and Gynecology MFM, identified the potential of exploring maternal supplementation to positively impact and benefit gut health, and called for further investigation.
Professor Paul Cotter, Principal Investigator at APC, said, "The low level of strain transfer detected in our initial study highlights that strain transfer can be used as a means of getting probiotic strains to the guts of infants through their administration to mothers. While the particular strain (Bifidobacterium breve 702258) did not transfer very efficiently, it did provide very valuable 'proof of concept data' prompting us to further explore the possibility of beneficial supplement intervention during pregnancy and transfer to the child."
Professor Fionnuala McAuliffe, UCD full academic professor of obstetrics and gynecology at National Maternity Hospital Dublin and Director of the UCD Perinatal Research Center, said, "This is significant and exciting research. By using our combined approach of metagenomic sequencing with culture and whole genome sequencing, our study has provided the most in-depth investigation to date of the transmission of Bifidobacterium from mother to infant. We have identified key clinical factors associated with beneficial transfer of Bifidobacterium from mother to baby such as spontaneous vaginal birth."
Professor Douwe van Sinderen, principal investigator at APC, commented, "This is a fascinating and highly collaborative project involving a number of institutions from academia, medicine and industry. Our research shows that maternal-to-infant transmission is a common phenomenon and established that many bifidobacteria naturally present in mothers transferred from mom to baby very effectively and transmission is strongly influenced by external factors including mode of delivery and antibiotics. This will benefit from further research and may point to a role for probiotic supplements during pregnancy, for targeted infant microbiome and health benefits."
More information: Conor Feehily et al, Detailed mapping of Bifidobacterium strain transmission from mother to infant via a dual culture-based and metagenomic approach, Nature Communications (2023). DOI: 10.1038/s41467-023-38694-0