This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Noninvasive modulation of brain hemodynamic and metabolic states by near-infrared light
The human brain is a dominant site for energy metabolism in the body. The intrinsic contraction and relaxation of the vascular walls, also known as "vasomotion," is responsible for metabolic and hemodynamic activity in the human brain. Disturbances in vasomotion are associated with neurological conditions, such as atherosclerosis, Alzheimer's disease, Parkinson's disease, and stroke.
A better understanding of the mechanisms underlying the hemodynamic and metabolic activities in human cerebral metabolism can help advance the application of neuromodulation in improving cognitive functioning in people with or without neurological disorders.
Transcranial photobiomodulation (tPBM) is an emerging form of light therapy that uses low intensity lasers or LEDs emitting near-infrared light to stimulate the brain and enhance its activity. A promising approach, tPBM is currently being explored as a potential therapy for various neurological conditions. However, it is still in its early stages of development, with researchers working on optimizing factors such as light intensity, wavelength, and treatment duration to achieve the best possible outcome.
Professor Hanli Liu and her colleagues from the University of Texas at Arlington, U.S., have been examining the specific effects of tPBM on the hemodynamic (relating to blood flow) and metabolic activity of the prefrontal cortex, a region of the brain involved in cognitive function. In their recent study published in Neurophotonics, they took things forward by showing that the hemodynamic and metabolic activities of the resting prefrontal cortex are indeed significantly modulated by tPBM.
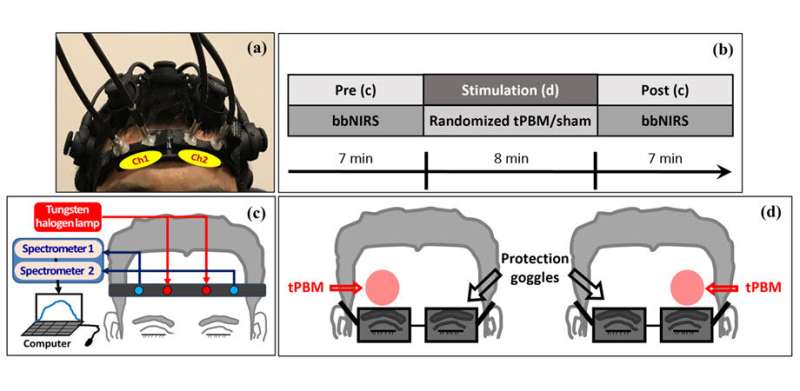
In this study, the researchers enrolled 26 healthy young adults to undergo tPBM sessions, during which they were subjected to five different stimulation conditions. Each participant received an 8-minute treatment with 800-nm and 850-nm light on the right forehead (R800, R850) and 800-nm light on the left forehead (L800). They also received two sham (placebo) interventions on the left and right forehead for control analysis. Laser-protection goggles were provided to protect the participants' eyes during the treatment.
Right before and after the 8-minute tPBM treatment, 2-channel broadband near-infrared spectroscopy (2-bbNIRS) detecting probes were placed on each side of the participant's foreheads. This setup helped the researchers capture and measure the effects of the treatment and changes in brain activity before and after the stimulation based on the absorption and scattering properties of the cerebral tissue. The measured data were then converted into signals to represent the hemodynamic and metabolic activities in different brain regions.
The researchers observed significant differences in the brain's response across infra-slow oscillation (ISO) bands—frequency ranges of rhythmic oscillations in blood vessels that occur independently of heartbeat—following tPBM treatment. Different oscillations of the ISO band, categorized as endogenic (E), myogenic (M), and neurogenic (N) components, represent the inner endothelial layer and outer smooth-muscle layer of blood vessels, as well as nerve-related elements within tissues, respectively.
In general, vasomotions in different regions of the brain, such as the bilateral forehead, are synchronized because of the unified heartbeat or cardiac motion. The researchers found that all three tPBM conditions resulted in reduction of synchronization between bilateral metabolism in the N band and bilateral hemodynamic activities in the M band of ISO. However, R800 significantly increased the bilateral hemodynamic and metabolic connectivity or synchronization in the E band.
Upon examining the coupling between hemodynamic and metabolic activities on each side of the prefrontal cortex, they further found that all three stimulation conditions increased hemodynamic-metabolic coupling in the E band only on the tPBM side, not on the opposite side. Also, both R800 and L800 led to enhanced hemodynamic-metabolic coupling in the M band also on the stimulation side.
Overall, these findings highlight that responses of the hemodynamic and metabolic activities of the prefrontal cortex to tPBM are dependent on the light wavelength as well as the site of stimulation application. Moreover, the endothelial and smooth-muscle cells in the blood vessels react differently to tPBM based on their vasomotion rhythms.
While further research is needed to support the application of tPBM in treating neurological conditions, these findings provide key scientific evidence that tPBM has the therapeutic potential to modulate brain connectivity across the forehead, and metabolic-hemodynamic coupling on the stimulation side.
More information: Sadra Shahdadian et al, Site-specific effects of 800- and 850-nm forehead transcranial photobiomodulation on prefrontal bilateral connectivity and unilateral coupling in young adults, Neurophotonics (2023). DOI: 10.1117/1.NPh.10.2.025012