This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Tracking somatostatin for early diabetes detection
Patients with diabetes have high blood glucose levels either because their pancreas does not produce sufficient insulin, or their body cells do not respond to signals from insulin that tell them to use up glucose. Current treatments, therefore, rely on supplying insulin bolus to such patients. However, recent studies have shown that fluctuations in the levels of other hormones such as somatostatin—also secreted by the pancreas—may be involved in the development of diabetes, something that has largely been overlooked so far.
"Changes in somatostatin secretion can be one of the first signs of diabetes," explains Nikhil Gandasi, Assistant Professor at the Department of Developmental Biology and Genetics (DBG), Indian Institute of Science (IISc). In such a scenario, detecting the levels of somatostatin can therefore potentially help detect diabetes earlier.
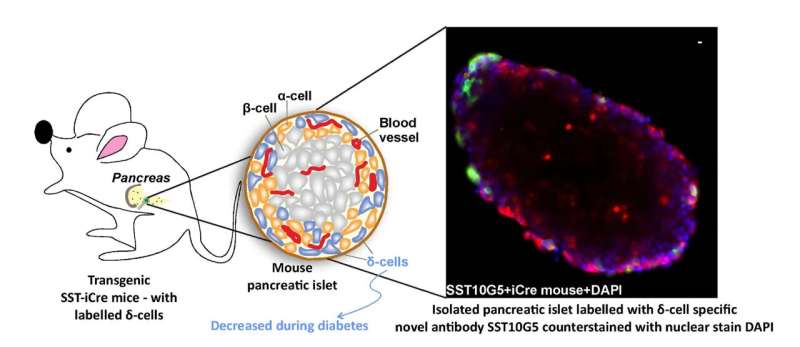
In a study published in the International Journal of Molecular Sciences, Gandasi's team, along with collaborators at the University of Gothenburg, Sweden, report the development of a novel assay that can detect secreted somatostatin. Somatostatin is secreted by specific cells of the pancreas, called delta cells. It is a master regulator of insulin and glucagon—another hormone that works hand-in-hand with insulin to maintain blood sugar levels.
The kit works like the standard Enzyme-Linked Immunosorbent Assay or ELISA which uses antibody-coated plates to identify the presence of antigens in a sample, similar to the COVID-19 rapid antigen test. The team used artificially synthesized somatostatin to test its binding against several commercially available antibodies, in order to identify the one that bound to it most efficiently, which they then used to develop the assay.
Using this assay, the researchers were able to detect the levels of somatostatin in pancreatic cells extracted from both mice and humans. In addition, they were able to measure the number of the delta cells that produce somatostatin in both human healthy and diabetic donor tissues. What they found was that the number of delta cells was drastically reduced in diabetic patients.
"There are less number of delta cells in the diabetic patient, therefore we see less secretion of somatostatin," says first author Lakshmi Kothegala, senior scientist at the University of Gothenburg, Sweden, and visiting scientist at IISc.
Currently, researchers rely on Radioimmunoassay (RIA) to detect somatostatin levels. But RIA uses radioactive materials and needs to be carried out under stringent safety conditions. Besides, RIA takes at least three days to complete, and can only be carried out in specialized labs, says Caroline Miranda, another first author and post-doctoral fellow at the University of Gothenburg.
"Having a fully functional ELISA will mean a more practical method, with faster results," she adds. The newly developed kit also requires only one-tenth of the volume of blood plasma sample needed for RIA.
The researchers are currently working with an industry collaborator to develop the kit into a simple handheld device that can eventually be mass-produced.
More information: Lakshmi Kothegala et al, Somatostatin Containing δ-Cell Number Is Reduced in Type-2 Diabetes, International Journal of Molecular Sciences (2023). DOI: 10.3390/ijms24043449