This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Researchers publish consensus statement on patient selection and appropriate use of Lu-177 PSMA-617 radionuclide therapy
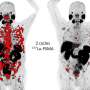
The Society of Nuclear Medicine and Molecular Imaging (SNMMI) has issued a new consensus statement to provide standardized guidance for the selection and management of metastatic castrate-resistant prostate cancer (mCRPC) patients being treated with 177Lu-PSMA radionuclide therapy.
The statement, published in The Journal of Nuclear Medicine, also reviews current clinical struggles physicians face during treatment with 177Lu-PSMA-617 radionuclide therapy.
Recently, the U.S. Food and Drug Administration approved 177Lu-PSMA-617 for the treatment of men with mCRPC after progressing on taxane-based chemotherapy and at least one line of androgen receptor pathway inhibitors (ARPI). To develop the patient selection criteria and determine appropriate use scenarios, SNMMI assembled an autonomous work group to represent a multidisciplinary panel of health care providers with substantive knowledge in the use of nuclear medicine in 177Lu-PSMA-617 radionuclide therapy. The work group conducted a review of the four prospective phase 2 and phase 3 trials that used 177Lu-PSMA-617 registered on clinicaltrials.gov to develop consensus recommendations.
In terms of patient selection, work group members agreed that PSMA PET should be performed within three months of treatment or since progression on the last therapy to ensure that the current disease state is represented. If disease progression or intervening therapy is evident, PSMA PET should be repeated. Either 18F-DCFPyL or 68Ga-PSMA-11 can be used for PSMA PET imaging, and 18F-FDG PET is not required as a standard patient selection tool. In addition, patients should be imaged with either contrast-enhanced CT or MRI to identify potential PSMA-negative disease. Baseline requirements for renal and bone marrow functions were also noted.
Treatment of mCRPC after chemotherapy and ARPI was considered appropriate, while treatment of mCRPC after ARPI and before chemotherapy was considered rarely appropriate. The working group also agreed that treatment of patients with metastatic castration-sensitive prostate cancer was rarely appropriate.
Furthermore, the consensus statement addressed current clinical struggles that physicians face when administering 177Lu-PSMA-617 radionuclide therapy. These include the role of androgen receptor-targeted therapies, the role of 223Ra, treatment-related toxicities, when to consider cessation of treatment, exceptional responders and restarting treatment, and imaging during treatment.
"With the approval of 177Lu-PSMA-617, a new class of therapeutics is available to patients with prostate cancer. We look forward to the potential use of PSMA radionuclide therapy in pre-chemotherapy mCRPC or other settings pending the full results of ongoing trials," noted the authors.
More information: Thomas A. Hope et al, SNMMI Consensus Statement on Patient Selection and Appropriate Use of177Lu-PSMA-617 Radionuclide Therapy, Journal of Nuclear Medicine (2023). DOI: 10.2967/jnumed.123.265952