This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Reimagining preclinical trials: Marmosets as model systems for the study of Alzheimer's disease
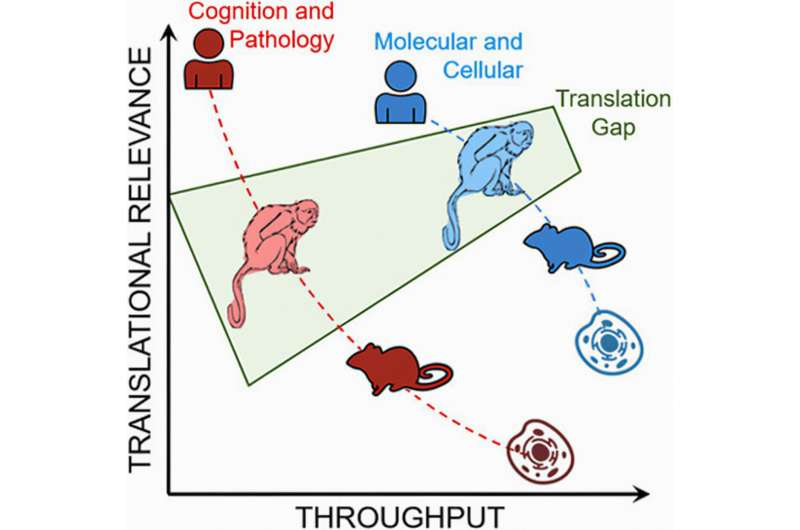
To reimagine existing preclinical trials for Alzheimer's disease, University of Pittsburgh School of Medicine neuroscientists created the first non-human primate model of hereditary Alzheimer's in marmoset monkeys, outlining their approach in Alzheimer's & Dementia: Translational Research & Clinical Interventions.
Researchers are now working on characterizing and validating genetic, molecular, functional and cognitive features of aging and Alzheimer's disease in marmosets that harbor mutations in the same gene that is linked to early-onset disease in humans. Scientists hope to accelerate the pace of the drug discovery pipeline and rebuild the foundation for future translational studies while overcoming limitations inherent to existing preclinical models.
"We are ambitious about finding a cure for Alzheimer's disease," said senior author Afonso Silva, Ph.D., professor of neurobiology at Pitt. "We are establishing a process for rigorous, minimally invasive standardized testing of the marmoset model of Alzheimer's disease and openly sharing data."
Marmoset families are better matched to mimic the genetically diverse human population than a colony of inbred rodents. And because marmosets' life spans are shorter than those of other non-human primates, researchers can comprehensively study their aging within a relatively short period of time.
If allowed to age naturally, marmosets will spontaneously develop aggregates of toxic amyloid beta and tau indicative of Alzheimer's-like pathology in the brain. To create marmosets with inheritable predisposition to Alzheimer's disease, researchers introduced a series of mutations in the PSEN1 gene using the Crispr/Cas9 gene engineering system. These same mutations cause early onset Alzheimer's disease in humans.
Presenilin-1, the protein encoded by PSEN1, plays a key role in generating amyloid tangles, and, just like human patients, marmosets with a mutation in the PSEN1 gene start developing Alzheimer's-like pathologies during adolescence.
In establishing the model, the team is applying a bench-to-bedside approach to marmosets as though they were human patients. To characterize and validate the new model, researchers are employing a battery of non-invasive tests, including behavioral studies, longitudinal analysis of blood biomarkers, and regular PET scans to assess brain function and pathological changes in the brain tissue.
The tests are designed to map out and compare the aging trajectory between healthy controls and animals genetically predisposed to early-onset Alzheimer's and correlate progressive changes in the levels of amyloid and tau to changes in cognition.
Researchers also plan to look into other factors that accompany disease progression, including brain-blood barrier permeability, vascular stiffness and metabolism, as well as analyze changes in gene expression profiles over time by regularly sampling skin cells.
"We need new models to understand underlying biological processes behind normal and pathological aging," said lead and corresponding author Stacey Rizzo, Ph.D., associate professor of neurobiology and deputy director for preclinical research at Pitt's Aging Institute. "Tracking the animals from birth in a well-controlled way would allow us to hypothesize on how molecular and genetic changes translate to pathophysiological consequences in the brain and devise ways to stop them from getting to the point of no return."
More information: Stacey J. Sukoff Rizzo et al, Bridging the rodent to human translational gap: Marmosets as model systems for the study of Alzheimer's disease, Alzheimer's & Dementia: Translational Research & Clinical Interventions (2023). DOI: 10.1002/trc2.12417





















