This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers uncover why a gene mutant causes young children to have strokes
A discovery of a mutation in the gene ACTA2 has given researchers, led by Dianna Milewicz, MD, Ph.D., with UTHealth Houston, insight into understanding the cause of a rare and progressive problem with arteries in the brain and a cause of strokes in young children, called moyamoya disease.
The findings were published today in Nature Cardiovascular Research.
Moyamoya disease is a condition where the arteries going through the neck and into the brain become blocked right when the arteries enter the brain. Moyamoya disease can lead to strokes and seizures. Children only a few months old can suffer a stroke due to the disease. Current treatments are limited to medications to reduce the risk of stroke and surgery to open or bypass blocked arteries.
"This disease is one of the major causes of stroke in children and nobody knows why this happens or why these arteries get clogged," said Milewicz, senior author of the study and professor and director of the Division of Medical Genetics at McGovern Medical School at UTHealth Houston. "These children don't smoke; they don't have hypertension and they don't have any of the other usual risk factors that cause strokes in adults."
Researchers identified that a change in the gene called ACTA2 caused children to have moyamoya disease and strokes starting shortly after birth, a condition called Smooth Muscle Dysfunction Syndrome, and have been working to figure out how and why this ACTA2 change causes moyamoya disease and strokes. Previous research led by Milewicz identified that ACTA2 mutations are the cause of Smooth Muscle Dysfunction Syndrome. In addition to moyamoya disease, this condition causes dysfunction of smooth muscle cells throughout the body.
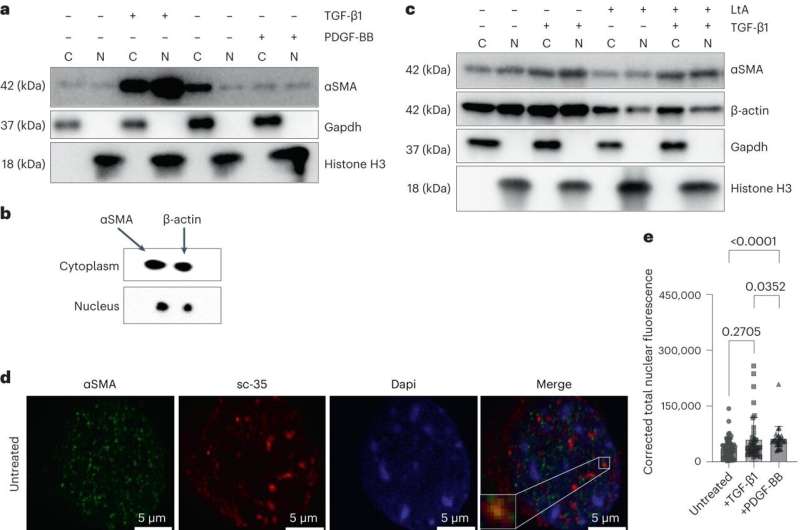
ACTA2 is found in the smooth muscle cells, which line the arteries and allow them to contract to control blood pressure and flow. Using model systems, including cells from patients with the ACTA2 variant that causes moyamoya disease, Milewicz and her team found that a mutation in ACTA2 causes the cells in the walls of the arteries in the brain to not differentiate properly, an essential component of vascular development.
"We found a new job that the ACTA2 protein is supposed to do that the mutant version cannot: to help make differentiated smooth muscle cells that stay in the blood vessel and contract to regulate blood pressure," said Callie Kwartler, Ph.D., first author of the study and assistant professor in the Division of Medical Genetics at McGovern Medical School.
The result is that the cells with the ACTA2 variant continue to grow out of control and move into the inside of the artery, which may be the cause of blockages in the arteries.
"This is the first step into really understanding the cause of moyamoya disease," said Milewicz, the President George Bush Chair in Cardiovascular Medicine with McGovern Medical School. "This is a disorder that starts out in childhood, and children with Smooth Muscle Dysfunction Syndrome die from strokes. We are working to use the information to prevent strokes in these children."
Researchers will continue to focus on exploiting the mechanism of disease that they identified to find new treatment options for children with moyamoya disease.
More information: Callie S. Kwartler et al, Nuclear smooth muscle α-actin participates in vascular smooth muscle cell differentiation, Nature Cardiovascular Research (2023). DOI: 10.1038/s44161-023-00337-4