This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Q&A: Expert explains RSV prevention for vulnerable populations

Respiratory syncytial virus (RSV) is a common virus that seriously affects vulnerable populations such as infants, young children and older adults. Dr. Wasem Alsabbagh, associate professor at the University of Waterloo School of Pharmacy, answers frequently asked questions about RSV.
Why should I be concerned about RSV?
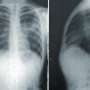
RSV is the leading cause of respiratory tract infections and hospitalizations among infants in Canada. About 80% of children who are hospitalized with bronchitis have RSV, while 20% of infants hospitalized with pneumonia have RSV.
What are the symptoms of RSV?
Patients with RSV present with fever, chills, difficulty sleeping and/or eating. Symptoms in infants range from nasal congestion, cough, and runny nose to severe symptoms of wheezing, shortness of breath, fast breathing and may lead to pneumonia or bronchitis. Parents are urged to seek medical care immediately when symptoms of severe RSV develop.
Who is most at risk for severe RSV?
Risk factors for severe RSV infection include premature birth, chronic lung illness of prematurity, congenital heart failure, neuromuscular impairment, immunodeficiency and Down syndrome. However, severe RSV can happen to any infant and almost 80% of hospitalizations in 2022 for RSV were among healthy infants.
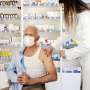
Can I get an RSV vaccine?
GSK's RSV vaccine (Arexvy) is approved by Health Canada for people aged 60 and older. There is no vaccine for children and infants yet.
Will there be an RSV vaccine for children and adults?
Ontario has started a publicly funded RSV vaccine program tailored and covered in full for older high-risk adults residing in long-term care homes, Elder Care Lodges and retirement homes licensed to provide dementia care services.
Can I get my flu shot, COVID-19 booster and RSV vaccine at the same time?
It is recommended that the GSK RSV vaccine not be routinely co-administered with COVID-19 or influenza vaccines, at least 14 days before or after receiving them. COVID-19 vaccines are recommended to be co-administered with the seasonal influenza vaccine.