This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Cell-type-specific genetic risk contributes to distinct stages of Alzheimer's disease progression, finds study
Developing treatments for Alzheimer's disease (AD) is difficult because complex underlying mechanisms drive different types of cells that may contribute to the disorder. Microglia and astrocytes, resident immune and support cells in the central nervous system, are known to exclusively express several genes linked to the risk of AD—particularly AD dementia.
However, it was previously unclear exactly how and when these genetic risk factors contributed to other, distinct stages of AD progression, such as the accumulation of amyloid-β plaques and tau tangles.
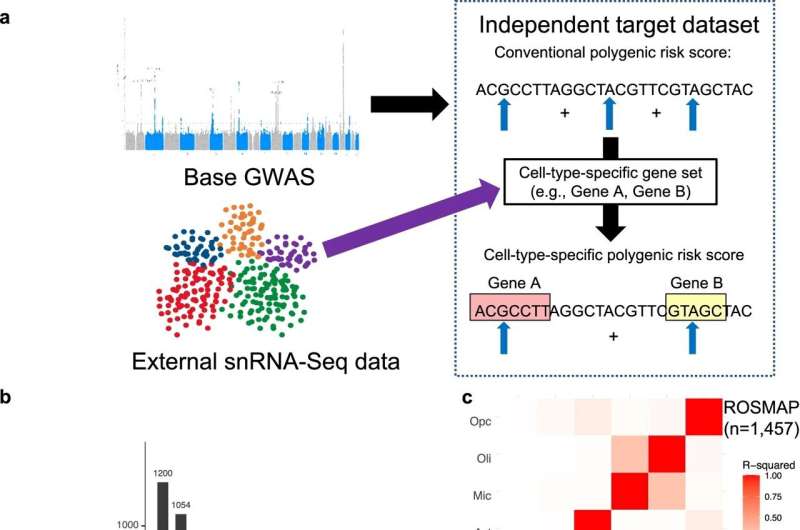
Researchers led by a team at Brigham and Women's Hospital, a founding member of the Mass General Brigham health care system, identified the impact of AD genetic risk specific to each major brain cell type on key disease processes. They implemented single nucleus RNA sequencing to calculate cell-type-specific AD polygenic risk scores from two large clinical research study datasets.
Using autopsy data spanning all stages of disease severity, along with independent neuroimaging data from asymptomatic, preclinical stages of AD, the investigators were able to characterize the contributions of cell-specific risk genes. Astrocyte-specific genetic risk contributed to earlier stages of disease progress, like amyloid-β accumulation, while microglia-specific risk played a part in later phases of plaque and tau tangle accumulation and cognitive decline.
"Our results provide human evidence for how genetic risk in specific brain cells affects AD processes, some even before the onset of clinical symptoms," said Hyun-Sik Yang, MD, of the Department of Neurology. "Future studies could extend our technique to other aspects of AD or even other diseases in order to help develop targeted treatments."
The research is published in the journal Nature Communications.
More information: Hyun-Sik Yang et al, Cell-type-specific Alzheimer's disease polygenic risk scores are associated with distinct disease processes in Alzheimer's disease, Nature Communications (2023). DOI: 10.1038/s41467-023-43132-2