This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Examining diabetes with a skin scanner and AI
Changes in small blood vessels are a common consequence of diabetes development. Researchers at the Technical University of Munich (TUM) and Helmholtz Munich have now developed a method that can be used to measure these microvascular changes in the skin—and thus assess the severity of the disease. To achieve this, they combine artificial intelligence (AI) and innovative high-resolution optoacoustic imaging technology. The work is published in the journal Nature Biomedical Engineering.
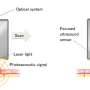
Optoacoustic imaging methods use light pulses to generate ultrasound inside tissue. The ultrasound waves generated are then recorded by sensors and converted to images. The signals are caused by tiny expansions and contractions of tissue that surround molecules that strongly absorb light. One such molecule is hemoglobin. Since hemoglobin is concentrated in blood vessels, optoacoustic imaging can produce detailed unique images of vessels in ways not possible by other non-invasive techniques.
The basic principles of optoacoustics, or photoacoustics, have been known for more than a century, but practical applications in medicine are fairly recent. Vasilis Ntziachristos is Professor of Biological Imaging at TUM and Director of the Institute of Biological and Medical Imaging and of the Bioengineering Center at Helmholtz Munich. Together with his team, he has developed a range of optoacoustic imaging methods, among them RSOM, short for raster-scan optoacoustic mesoscopy.
32 particularly significant changes
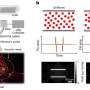
The researchers have now successfully employed RSOM to study the effects of diabetes on the human skin. Using RSOM images of the blood vessels in the legs of 75 diabetics and a control group, the researchers identified characteristics of diabetes using an AI algorithm.
They created a list of 32 particularly significant changes based on alterations of the skin microvasculature appearance. These included features such as the number of branches of the vessels or their diameter.
RSOM allows for quick measurement of vascular changes
The fact that small blood vessels in the skin of diabetics are altered is well-established based on biopsies, i.e. examinations of small parts excised from the skin. Biopsies, however, do not accurately represent the living conditions since they may deform the blood vessels. They are also invasive and not suited for observations over an extended period of time.
RSOM measurements, on the other hand, are not invasive, take less than a minute and do not rely on radiation or contrast agents. "Other optical methods do not achieve the depth or the detail reached by RSOM," says Angelos Karlas, lead clinician in the study.
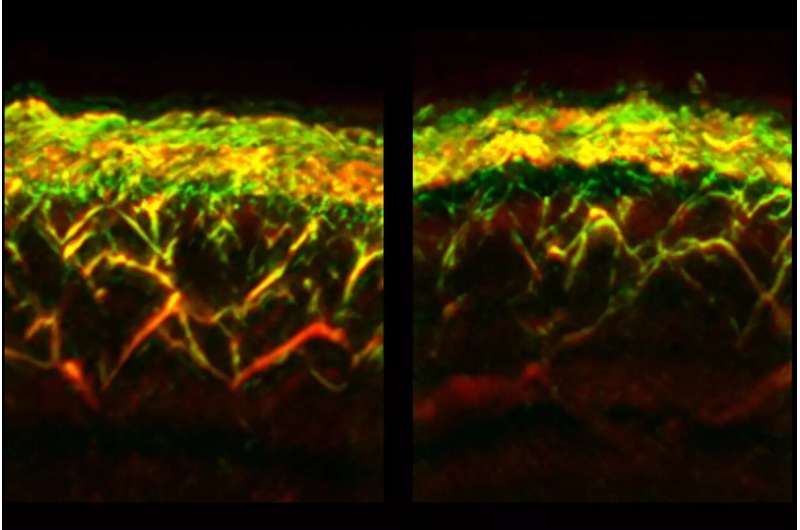
With a single RSOM measurement, data on different depths of the skin can be obtained simultaneously. This enabled the researchers to determine for the first time that diabetes affects vessels at different skin layers differently. For example, while the number of vessels and branches in the so-called dermal layer were reduced in diabetics, they were increased closer to the surface of the skin, in the so-called epidermal layer.
Assessing diabetes stage by combining skin features
All the 32 characteristics mentioned above are affected by the progression and severity of the disease. Yet, only when they are combined and a score is calculated, a link can be drawn time between the condition of the small blood vessels in the skin and the severity of diabetes. This is achieved for the first time in the current study.
"With RSOM, we can now quantitatively describe the effects of diabetes," says Vasilis Ntziachristos. "With the emerging ability to make RSOM portable and cost-effective, these findings open up a new way for continuous monitoring of the status of those affected—more than 400 million people worldwide. In the future, with fast and painless examinations, it would take just a few minutes to determine whether therapies are having an effect, even at home environments."
More information: Angelos Karlas et al, Dermal features derived from optoacoustic tomograms via machine learning correlate microangiopathy phenotypes with diabetes stage, Nature Biomedical Engineering (2023). DOI: 10.1038/s41551-023-01151-w