This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Risk model identifies metastatic cancer patients who may benefit from immune checkpoint inhibitors
Immune checkpoint inhibitors (ICIs) are used to treat a variety of different cancers. ICIs are antibodies that sensitize the body's own immune system to detect and destroy tumors. However, ICIs are not effective in all patients, and systemic cancer-related inflammation, for example, may influence the efficacy of ICIs.
A risk model developed by researchers can help to identify cancer patients who could benefit from immune checkpoint inhibitors. Published in BMC Cancer, the study was conducted at the University of Eastern Finland and Kuopio University Hospital.
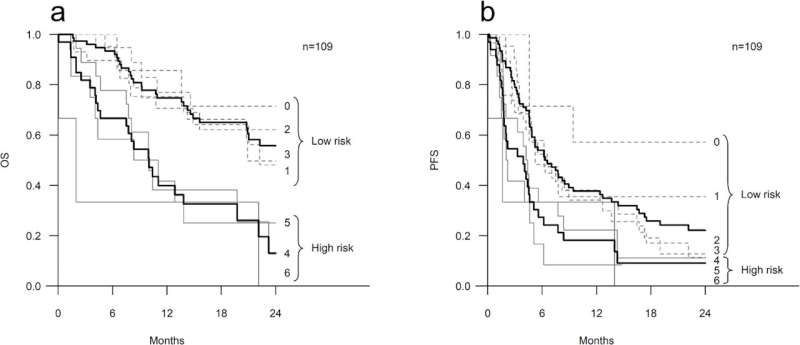
In the new study, six inflammation-related laboratory parameters were used to create a risk model that classified patients receiving ICIs into low-risk and high-risk groups.
"With a risk model that predicts treatment outcomes, treatment can be better targeted at patients who are more likely to benefit from it," says Senior Researcher Aino Rönkä of the University of Eastern Finland.
The study cohort consisted of patients receiving ICIs for metastatic cancers at Kuopio University Hospital Cancer Center. Patients were given a risk score of 0–6 based on the following inflammation-related laboratory parameters: elevated values of neutrophils, platelets, C-reactive protein (CRP), lactate dehydrogenase, erythrocyte sedimentation rate and the presence of anemia.
Based on their risk score, patients were classified into two groups: those with a risk score of 0–3 to the low-risk group indicative of a good prognosis, and those with a risk score of 4–6 to the high-risk group indicative of a poor prognosis.
In the low-risk group, 53.9% of patients responded to ICIs, whereas in the high-risk group the response rate was 30.3%. The median overall survival was 27.3 months in the low-risk group, and 10 months in the high-risk group. The risk model appeared to be functional in all the common cancer types studied, i.e., lung cancer, melanoma and renal cell carcinoma.
Relying on routine blood work, the risk scoring developed in the study is a practical predictive model that could easily be used in cancer patients' treatment assessment. Targeting ICIs to patients most likely to benefit from them increases the efficacy, safety and cost-effectiveness of treatment. However, the researchers point out that the model still needs to be validated in a prospective, multi-center setting.
More information: Satu Tiainen et al, A practical prognostic peripheral blood-based risk model for the evaluation of the likelihood of a response and survival of metastatic cancer patients treated with immune checkpoint inhibitors, BMC Cancer (2023). DOI: 10.1186/s12885-023-11699-0