This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Neuroscientific study paves the way for improving the diagnosis and treatment of Alzheimer's
The prevailing theory on the causes of Alzheimer's disease indicates that they are related to the abnormal accumulation of two proteins, amyloid-beta (Aβ) and tau, in neuronal tissues, but there are still many unknowns concerning the processes that trigger this neurodegenerative disease. To date, most scientific studies have analyzed the effect of one of these proteins, amyloid-beta, on neuronal dynamics, but without looking in depth at its complex interaction with tau in patients with Alzheimer's.
In this regard, recent neuroscientific research involving the UPF Center for Brain and Cognition (CBC) provides interesting developments on the dynamics and complex interaction of these two proteins in the brains of people with Alzheimer's. Their results pave the way for future research to design new biomarkers to improve its diagnostic processes and therapies for its treatment.
The results of this research are presented in an article published recently in the journal Alzheimer's Research & Therapy.
The main author of the article, Gustavo Patow, is linked to the Visualization, Virtual Reality, and Graphics Interaction (ViRVIG) research group of the University of Girona and to the Computational Neuroscience group of the CBC at UPF. Gustavo Deco (ICREA), director of the Computational Neuroscience group at the CBC, is also a co-author of the article.
The other co-authors, Leon Stefanovski, Petra Ritter and Xenia Koveleva, are linked to various universities and medical and research centers in Germany.
Gustavo Patow (ViRVIG-UdG and CBC-UPF) says, "Alzheimer's is a neurodegenerative disease that progressively degrades the brain to the point of destroying its normal functioning. Computational neuroscience methods, in particular simulation-based models, enable studying the evolution of the disease in ways that cannot be applied to real patients.
He clarifies that these "in-silico" simulations allow for formulating and testing hypotheses and provide information and, occasionally, new knowledge about the disease. "We have been able to determine the relative importance of the two proteins, which are believed to be the cause of Alzheimer's, throughout the evolution of the disease, which allows us to generate valuable information that helps with its diagnosis, since the study provides us with precise information about the patient's status based on their functional scanners, the so-called 'biomarkers.'"
Gustavo Deco (CBC-UPF and ICREA) adds, "The mathematical and computational tools, deeply rooted in our medical and biological knowledge of Alzheimer's, open new doors for the study of the evolution of the disease and, above all, to analyze possible strategies to combat its effects and, hopefully, slow its progress."
"Our group is working intensively on these types of predictive models that allow us to work towards this ideal, with the ultimate goal of fighting this terrible disease. The results of this work are a promising first step in this direction."
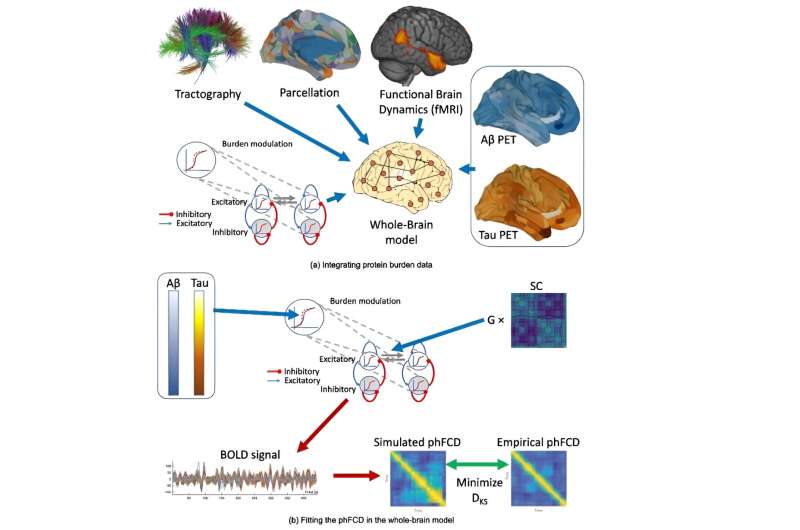
In this study, whole-brain modeling techniques have been used that allow us to understand the mechanisms underlying the global dynamics of the different brain regions in their different states (at rest, performing cognitive tasks...), both in healthy subjects and in people with the disease, by means of mathematical tools and operations.
Based on these techniques, the impact has been studied of the concentration of the two proteins (amyloid-beta and tau) on the behavioral dynamics of different brain regions of people with Alzheimer's disease. It was possible to ascertain the impact of each protein individually and combined with the other and to evaluate the relative weight of each of them in the abnormal brain activity of people with the disease.
The study concludes that in the early stages of Alzheimer's, the greatest impact of the disease is caused by a greater accumulation of amyloid-beta protein. In contrast, at a more advanced stage, this impact can be attributed to a higher concentration of tau protein. Both proteins alter the balance between the state of neuronal excitation and inhibition, which contributes to the cognitive deterioration of people suffering the disease.
Previous research into the disease had already detected that people with Alzheimer's suffer from an alteration of the levels of neuronal excitation and inhibition balance. Especially in the early stages of the disease, neuronal hyperexcitability occurs, which impairs cortical activity and thus contributes to cognitive decline. Until now, this imbalance had been demonstrated in animal studies and in post-mortem human samples, but evidence of this was lacking in humans living with the disease.
The main difficulty in achieving this is that neuronal excitation and inhibition activity cannot be directly measured using neuroimaging techniques. However, the whole-brain modeling techniques used in this research have indeed managed to measure the impact of the two proteins on the neuronal excitation/inhibition imbalance, using mathematical and computational simulation tools.
This study used empirical data obtained from the Alzheimer's Disease Neuroimaging Initiative database, a longitudinal multi-site study designed to develop biomarkers for Alzheimer's disease. The data correspond to subjects with the disease aged between 55 and 90 years.
More information: Gustavo Patow et al, Whole-brain modeling of the differential influences of amyloid-beta and tau in Alzheimer's disease, Alzheimer's Research & Therapy (2023). DOI: 10.1186/s13195-023-01349-9