This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Investigational therapeutic shows promise in preclinical pancreatic cancer model
A new type of investigational therapeutic in development for pancreatic cancer has shown unprecedented tumor-fighting abilities in preclinical models of the disease, suggesting it has the potential to offer novel treatment options for nearly all pancreatic tumors, a comprehensive study has found.
The inhibitors in this new class of oral medications, being developed by Revolution Medicines Inc., target the oncogenic or active cancer-causing form of RAS proteins (such as KRAS, NRAS, and HRAS). These RAS "oncoproteins" drive up to a third of all human cancers.
The research findings—conducted by a consortium of academic researchers led by Columbia scientists and the scientific team at Revolution Medicines—were published in a paper appearing in Nature titled "Tumor-selective effects of active RAS inhibition in pancreatic ductal adenocarcinoma."
Currently the third leading cause of death from cancer, pancreatic cancer kills about 50,000 people annually in the United States alone. Despite decades of research, the disease continues to stymie drug developers and oncologists. What's especially frustrating is that scientists know exactly what causes most cases at the cellular level.
"For over four decades, we have known that there's one particular RAS protein, called KRAS, that's mutated and drives about 95% of all pancreatic ductal adenocarcinoma cases, and we've had no direct tools to attack it for most of that time," says Kenneth Olive, Ph.D., associate professor of medicine at Columbia University's Vagelos College of Physicians and Surgeons and Herbert Irving Comprehensive Cancer Center, one of the study's senior authors.
When the study's co-senior author, Mallika Singh, Ph.D., vice president for translational research at Revolution Medicines, told Olive the company had invented a class of inhibitors that had the potential to target all RAS mutations, he was incredulous. "My immediate reaction was skepticism," says Olive. "But I was curious, and we quickly established a collaboration."
Study results
Preclinical studies soon launched in the Olive lab at Columbia, led by Urszula Wasko, a Ph.D. student in the molecular pharmacology graduate program. Early pilot experiments with RMC-7977 were remarkably effective.
"We immediately knew we were working with something entirely different," says Olive. At the same time, Olive and Revolution Medicines worked to bring together pancreatic cancer experts from other academic institutions, including the University of Pennsylvania, Dana-Farber Cancer Institute, University of North Carolina at Chapel Hill, and Memorial Sloan Kettering.
"Rather than compete against one another, we established a consortium and agreed to share data in real time. That was transformative," says Olive.
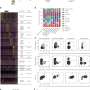
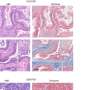
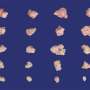
Pancreatic cancer researchers have developed many different preclinical models of the disease over the years, each with its own strengths and weaknesses. Rather than pick one, the expanded team tested RMC-7977 in all of them.
"By unleashing a consortium of scientists on this problem, we were able to examine active RAS inhibition in every major class of model for pancreatic cancer, and this inhibitor performed really well in all," says Olive.
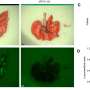
The preclinical tumor model Olive's lab has long favored is widely recognized for its broad resistance to treatment. "RMC-7977 as a single agent outperformed the best combination regimen that has ever been reported in the literature in that model system," he says, adding that it's the first time he's ever seen tumors routinely get smaller in those systems. Other models the consortium tested yielded similar results.
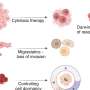
Because RMC-7977 also inhibits wild-type RAS proteins essential to the health of many normal cells, the scientists also carefully examined normal tissues in the treated animals. This work showed that tumor cells are uniquely sensitive to the inhibitor, while the impact in normal cells was minimal.
Though the initial responses in preclinical tumor models to the inhibitor were impressive, Olive hastens to point out that the tumors were not eliminated.
"In almost every case, the tumor came back," he says. In tissue culture, the investigators identified another oncogene, called MYC, that was altered in most of the resistant tumors, then developed a combination treatment that was effective against tumor cells that had developed resistance to the active RAS inhibitor. Those results suggest a combinatorial approach that is worth exploring in patients in the future.
In a field with a long history of failed drug development efforts, the new results are cause for optimism, Olive says. "I've been working on pancreatic cancer for almost 20 years, and I've never seen preclinical results like these. I think there is a real chance this approach will help change the standard of care for pancreatic cancer patients, but only clinical trials can determine that. I'm excited that Columbia is one of many institutions participating in the clinical development of these new agents."
More information: Kenneth Olive, Tumor-selective activity of RAS-GTP inhibition in pancreatic cancer, Nature (2024). DOI: 10.1038/s41586-024-07379-z. www.nature.com/articles/s41586-024-07379-z