I'm pregnant. Do I need a multivitamin?
Growing a healthy baby relies on getting enough nutrients while pregnant.
2 hours ago
0
0
Growing a healthy baby relies on getting enough nutrients while pregnant.
2 hours ago
0
0

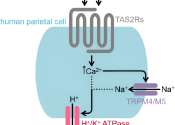
In the stomach, parietal cells are responsible for acid production. They react not only to the body's own messenger molecules, but also to bitter-tasting food constituents such as caffeine.
May 14, 2024
0
0
How did your heart form? What triggered your first heartbeat? To this day, the mechanisms of human heart development remain elusive.
May 13, 2024
0
0

Common conditions such as indigestion and heartburn as well as peptic ulcers, autoimmune gastritis and stomach and esophageal cancers have one thing in common—they involve disruptions of the normal activity of parietal ...
May 10, 2024
0
26

New research presented at the European Congress on Obesity (ECO) in Venice, Italy (12–15 May), and published in the International Journal of Obesity, could help explain why type 2 diabetes is more common in men than in ...
May 10, 2024
0
2

You wake up, stagger to the bathroom and gaze into the mirror. No, you're not imagining it. You've developed face wrinkles overnight. They're sleep wrinkles.
May 10, 2024
0
0

Researchers at MIT, Brigham and Women's Hospital, and Harvard Medical School have developed a potential new treatment for alopecia areata, an autoimmune disorder that causes hair loss and affects people of all ages, including ...
May 9, 2024
0
97

A group of researchers at the University of California San Diego School of Medicine have led an investigation that offers new insight into the causes of spina bifida, the most common structural disorder of the human nervous ...
May 8, 2024
0
12
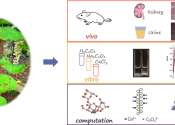
In the Dai minority region of China, the Aspidopterys obcordata (Hei Gai Guan) has been used as a fork medicine for the treatment of urinary tract infections, cystitis, and urinary tract stones. Previous studies showed that ...
May 8, 2024
0
0
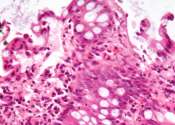
Inulin, a type of fiber found in certain plant-based foods and fiber supplements, causes inflammation in the gut and exacerbates inflammatory bowel disease in a preclinical model, according to a new study by Weill Cornell ...
May 3, 2024
4
329
An acid (from the Latin acidus/acēre meaning sour) is a substance which reacts with a base. Commonly, acids can be identified as tasting sour, reacting with metals such as calcium, and bases like sodium carbonate. Aqueous acids have a pH of less than 7, where an acid of lower pH is typically stronger, and turn blue litmus paper red. Chemicals or substances having the property of an acid are said to be acidic.
Common examples of acids include acetic acid (in vinegar), sulfuric acid (used in car batteries), and tartaric acid (used in baking). As these three examples show, acids can be solutions, liquids, or solids. Gases such as hydrogen chloride can be acids as well. Strong acids and some concentrated weak acids are corrosive, but there are exceptions such as carboranes and boric acid.
There are three common definitions for acids: the Arrhenius definition, the Brønsted-Lowry definition, and the Lewis definition. The Arrhenius definition states that acids are substances which increase the concentration of hydronium ions (H3O+) in solution. The Brønsted-Lowry definition is an expansion: an acid is a substance which can act as a proton donor. Most acids encountered in everyday life are aqueous solutions, or can be dissolved in water, and these two definitions are most relevant. The reason why pHs of acids are less than 7 is that the concentration of hydronium ions is greater than 10−7 moles per liter. Since pH is defined as the negative logarithm of the concentration of hydronium ions, acids thus have pHs of less than 7. By the Brønsted-Lowry definition, any compound which can easily be deprotonated can be considered an acid. Examples include alcohols and amines which contain O-H or N-H fragments.
In chemistry, the Lewis definition of acidity is frequently encountered. Lewis acids are electron-pair acceptors. Examples of Lewis acids include all metal cations, and electron-deficient molecules such as boron trifluoride and aluminium trichloride. Hydronium ions are acids according to all three definitions. Interestingly, although alcohols and amines can be Brønsted-Lowry acids as mentioned above, they can also function as Lewis bases due to the lone pairs of electrons on their oxygen and nitrogen atoms.
This text uses material from Wikipedia, licensed under CC BY-SA