Ultra short telomeres linked to osteoarthritis
Telomeres, the very ends of chromosomes, become shorter as we age. When a cell divides it first duplicates its DNA and, because the DNA replication machinery fails to get all the way to the end, with each successive cell division a little bit more is missed. New research published in BioMed Central's open access journal Arthritis Research & Therapy shows that cells from osteoarthritic knees have abnormally shortened telomeres and that the percentage of cells with ultra short telomeres increases the closer to the damaged region within the joint.
While the shortening of telomeres is an unavoidable side effect of getting older, telomeres can also shorten as a result of sudden cell damage, including oxidative damage. Abnormally short telomeres have been found in some types of cancer, possibly because of the rapid cell division the cells are forced to undergo.
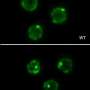
There has been some evidence from preliminary work done on cultured cells that the average telomere length is also reduced in osteoarthritis (OA). A team of researchers from Denmark used newly developed technology (Universal single telomere length assay) to look in detail at the telomeres of cells taken from the knees of people who had undergone joint replacement surgery. Their results showed that average telomere length was, as expected, shortened in OA, but that also 'ultra short' telomeres, thought to be due to oxidative stress, were even more strongly associated with OA.
Maria Harbo who led this research explained, "We see both a reduced mean telomere length and an increase in the number of cells with ultra short telomeres associated with increased severity of OA, proximity to the most damaged section of the joint, and with senescence. Senescence can be most simply explained as biological aging and senescent cartilage within joints is unable to repair itself properly."
She continued, "The telomere story shows us that there are, in theory, two processes going on in OA. Age-related shortening of telomeres, which leads to the inability of cells to continue dividing and so to cell senescence, and ultra short telomeres, probably caused by compression stress during use, which lead to senescence and failure of the joint to repair itself. We believe the second situation to be the most important in OA. The damaged cartilage could add to the mechanical stress within the joint and so cause a feedback cycle driving the progression of the disease."