Implant to replace defective venous valve
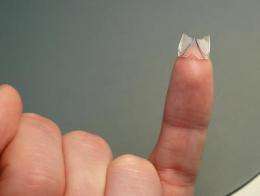
If heart valves don't close properly, they are replaced. Conventional treatment of venous valve failure, however, has up to now always and exclusively been via medication. In future, an implant will assume the function of damaged valves -- and a new dispensing tool means these prostheses can be made using an automated process.
It's one of the most commonly occurring medical conditions – chronic venous in-sufficiency (CVI). Almost ten million German citizens suffer from weak veins that require treatment, with twice as many women being affected as men. The cause of this widespread condition is restricted functioning of the venous valves in the legs. If the venous valve is no longer able to close properly, blood will observe the laws of gravity in between heartbeats and flow down to collect in the legs. This leads to edemas, and can cause open ulcers in particularly severe cases. CVI is usually treated with anti-inflammatory drugs and diuretics; as yet there is no globally available venous valve implant that can be used to treat the illness. This is something that researchers from the Fraunhofer Institute for Manufacturing Engineering and Automation IPA in Stuttgart are setting out to change: In close collaboration with four industrial partners and Helmholtz-Institute for Biomedical Engineering of RWTH Aachen University, they have developed an automated production facility that can make venous valve prostheses from polycarbonate-urethane (PCU), a plastic. The project was sponsored by the German Federal Ministry of Economics and Technology BMWI.
The centerpiece of the facility is a 3D droplet dispensing tool which enables the researchers to precisely apply a particular polymer onto freeform surfaces and at the same time combine various grades of polymer hardness, called Shore hardnesses. "3D droplet dispensing technology is an additive procedure that allows three-dimensional geometries to be created layer by layer using a polymer", explains Dr. Oliver Schwarz, group manager at the IPA. The scientists use PCU because it is particularly strong and flexible, while another useful property of the material is that it is easy to sew into surrounding tissue. PCU structures can be made in very thin layers, which is ideal when replacing wafer-thin atrioventricular valves. "By using PCU in combination with our 3D dispensing kinematics, we can achieve seamless transitions within the material between six different grades of elasticity and hardness – without any breaking points whatsoever. This technique mirrors the design of highly stressed structures in nature. It can't be done using injection molding", says Schwarz.
But how does the PCU become a venous valve prosthesis? Initially, the polymers are dissolved in a solvent and deposited onto a venous valve prosthetic mold one droplet at a time, using the dispensing tool. The system is accurate to within 25 micrometers, and can deliver up to 100 droplets per second, each with a volume of 2 to 60 nanoliters. A six-axis kinematic system positions the piezo feeder precisely above the mold. Once it is fully coated with droplets, the mold is bathed in a warm stream of nitrogen. This causes the solvent to evaporate, leaving the polymer behind. Further layers are applied by repeating the dispensing process, and in the end the polymer prosthesis can simply be peeled from the mold. Doctors can take the finished replacement valves and implant them into the veins of the leg via a catheter passed through the skin.
The production facility comprises numerous other components besides the dispenser. The IPA experts are responsible for, amongst other things, the filling and monitoring system, the drying facilities, the entire clean-room box and the control mechanism for the six-axis kinematic system. "We have successfully managed to re-program the Beckhoff control system normally used with milling machines in such a way that it can now be used with additive processes," Schwarz is happy to report. The solution they have come up with will soon see the researcher and his team in a position to produce thin-walled, highly durable implants such as heart valves or intervertebral disks.
The IPA scientists will be presenting a prototype of their 3D droplet dispenser at the MEDTEC Europe 2012 trade fair from March 13 – 15 in Stuttgart (Hall 6, Booth 6211).
















