April 9, 2012 feature
Dreamless nights: Brain activity during nonrapid eye movement sleep
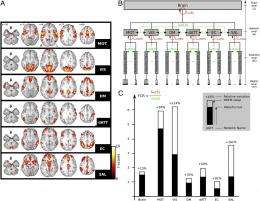
(Medical Xpress) -- The link between dreaming and rapid eye movement (REM) sleep are well understood – but the fact that consciousness is reduced during nonrapid eye movement (NREM) sleep is not. Recently, scientists in the Cyclotron Research Centre at the University of Liège, in Liège, Belgium, and the Institut National de la Santé et de la Recherche Médicale at the Université Pierre et Marie Curie in Paris, and the Functional Neuroimaging Unit at the Montreal Geriatrics Institute, investigated NREM sleep with the hypothesis that this phenomenon is associated with increased modularity of the brain’s functional activity during these periods. Using functional clustering – which estimates how integration is hierarchically organized within and across the constituent parts of a system they found that while in NREM sleep, hierarchically-organized large-scale neural networks were disaggregated into smaller independent modules. The researchers concluded that this difference could reduce the ability of the brain to integrate information, thereby accounting for the decreased consciousness experienced during NREM sleep.
Led by Pierre Maquet at the Cyclotron Research Centre and Habib Benali at the Institut National de la Santé et de la Recherche Médicale, the team faced a fundamental challenge in framing their research. Maquet first notes that there is currently no consensus on what consciousness really is, let alone how it arises.
“For many years,” he explains to Medical Xpress, “Giulio Tononi put forward the hypothesis that consciousness is related to the ability of the brain to integrate information. Our objective was simply to test this hypothesis, using novel tools allowing for the computation of information exchange within the brain and a set of EEG/fMRI data recorded in the same individuals during wakefulness and deep NREM sleep.” The latter state, he adds, is arguably the condition associated with the most reduce conscious content in normal human volunteers.
Maquet notes that the team used methods devised by Benali. “These allow us to measure the hierarchical organization of integration – i.e., information. The data itself,” he continues, “were acquired in Liège. Conducting simultaneous EEG/fMRI recordings in sleeping volunteers is not that easy.” Moreover, he notes, in practice, their findings are only one small step toward a better understanding of consciousness – and, for that matter, unconsciousness.
“The results were rather unexpected in that the amount of information exchanged in the brain actually increased during sleep. However, the patterns of exchange between regions were different than during wakefulness. Essentially, there was an increased information exchange within small clusters of mainly homologous brain areas whereas communication between clusters significantly decreased during sleep.” Thus, he points out, the data support their hypothesis.
The team has already defined the next steps in their research, says Maquet, who acknowledges that fMRI suffers from a rather sluggish signal. “The next step is to apply the methods to EEG, which has a much better time resolution.” He also states that it might it be possible to transition to in silico modeling, and that there are attempts in this direction in some laboratories.
A key advantage of the team’s approach was relying on functional clustering rather than so-called total integration in neural network analysis. “This is a big question,” states Maquet. “We don't know what is the information exchanged within clusters, and I don't see any technique that could currently answer this question in humans. More generally,” he adds, “it is thought that NREM sleep is regulated by the homeostasis of synaptic strength, and perhaps by neuronal energy metabolism.” These assumptions, he concludes, are being studied in animal models.
More information: Hierarchical clustering of brain activity during human nonrapid eye movement sleep, PNAS Published online before print March 26, 2012, doi: 10.1073/pnas.111113310
Copyright 2012 Phys.Org
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.