Electronic nose could be used to detect sleep apnoea
An electronic nose, used to detect the presence of molecules in the breath of a patient, could be used to diagnose obstructive sleep apnoea.
A new study, published online today ahead of print (25 October 2012) in the European Respiratory Journal, could make the diagnosis of the condition quick and inexpensive compared to current methods.
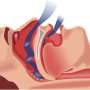
The gold standard used to identify sleep apnoea is an overnight sleep test. This is technically demanding, time-consuming and cost-intensive.
Electronic nose devices have been shown to distinguish between a number of diseases; they do this by analysing the pattern of volatile organic compounds in breath samples. This is the first study that has assessed whether the electronic nose could be used to confirm the presence of sleep apnoea.
Researchers analysed the breath of 40 sleep apnoea patients and 20 healthy controls. Sleep apnoea is associated with inflammation in the upper airways, which the researchers suggested could be detected by analysis of the exhaled breath of the patients. The study also aimed to assess whether the electronic nose could detect the effects of the standard treatment for sleep apnoea i.e. continuous positive airway pressure (CPAP).
The researchers performed questionnaires and sleep examinations to confirm sleep apnoea in 40 patients. They also collected throat washings from patients to measure any improvement in their condition following treatment with CPAP. The researchers then used a statistical analysis model to calculate the accuracy of the electronic nose.
The results found that the electronic nose could effectively diagnose sleep apnoea. The statistical analysis showed that sleep apnoea was detected with a sensitivity of 93%.
Lead author, Dr Timm Greulich, from the Marburg Hospital in Germany, said: "This is the first time an electronic nose has been tested in the setting of sleep apnoea diagnosis. The electronic nose could be useful in two ways. First, it can rule out the disease in a low prevalence population. Secondly, in a population with a high risk of sleep apnoea, the device could be used to help decide who would need to undergo an overnight sleep examination. Following these results, we foresee that the use of an electronic nose could reduce costs by more appropriately selecting patients who require the sleep examination."
European Respiratory Journal Associate Editor, Professor Ramon Farre, commented: "This is an interesting study, adding to a growing body of evidence demonstrating that electronic noses are a future potential tool for the detection of respiratory diseases. Although the results are encouraging, it is important to acknowledge the limitations in this kind of statistical analysis and it is not yet definite proof that this is an accurate diagnostic tool. It is, however, the first study to consider the application of an electronic nose for sleep apnoea and we look forward to seeing future research expanding on this important initial research."
More information: European Respiratory Journal DOI: 10.1183/09031936.000917.12