Genetic background check may explain why mutations produce different results

Two women have the same genetic mutation – an abnormal BRCA1 gene that puts them both at much higher-than-average risk for breast cancer – but only one woman develops the disease. Why? Michigan State University genetic scientists have begun to understand the mechanisms behind the phenomena.
"It's been known for a while that genetic mutations can modify each other," explained Ian Dworkin, MSU associate professor of zoology. "And we also know that the subtle differences in an individual's genome – what scientists call wild type genetic background—also affects how mutations are manifested. We wanted to know how common it was for wild type genetic background to alter the way genetic mutations interact with each other. No one has really looked at this in a systematic manner before."
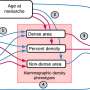
Using the fruit fly genome, Dworkin and Sudarshan Chari, zoology doctoral student, found that wild type genetic background affected the outcomes of interactions between genetic mutations about 75 percent of the time, which could have huge implications in how scientists construct genetic networks—maps of how genes interact with each other.
"It may be that some crucial portions of genetic networks are missing," Dworkin said. "It also seems that network descriptions are more fluid than we thought."
The research, "The conditional nature or genetic interactions: the consequences of wild-type backgrounds on mutational interactions in a genome-wide modifier screen," is published in the Aug. 1, 2013 issue of PLoS Genetics.
For their study, Dworkin and Chari looked at a fruit fly genetic mutation in the gene scalloped, which affects the size and shape of the flies' wings. They painstakingly went through the entire fly genome and crossed the scalloped mutation with another mutation that was in almost all the other DNA in the genome, one segment at a time, to see how often the genetic background affected the physical outcome of the scalloped mutation.
"We were surprised that the influence of wildtype genetic background on genetic interaction was so common," Dworkin said. "We knew it could happen, but no one had demonstrated that it was this prevalent. The broader implication is that even for diseases with a simple genetic basis, variation in the genome may matter for both understanding and treatment."
While the fruit fly and human genomes are different, the two species share a number of genes, with the fruit fly often having one copy of a gene while humans have multiple copies. The human version of the fruit fly scalloped gene is called TEF-1, which regulates tissue growth among other things. The flies have one copy of the scalloped gene while humans have multiple copies of the TEF genes.
So going back to the example of the two women with the same BRCA1 mutation, each woman's genetic background is likely influencing how the mutation is expressed, causing two very different disease outcomes.
The research also may help explain why some people benefit from a specific treatment for a disease, while others get no benefits or become resistant to a drug after a short time.
It's likely that most diseases with a suspected genetic component, such as cancer, asthma, or Parkinson's, involve reactions between more than one set of genes. For Dworkin and Chari, the next step is to start to tease apart the intricacies of what's happening.
"Is it just the two pairs of genes that are interacting?" Dworkin said. "Or is it that the two genes are interacting and then many other genes are modifying that reaction? This will help us understand how much complexity is involved."