August 16, 2013 report
Universal flu vaccine may be a step closer
(Medical Xpress)—A new study in the US suggests that boosting a certain group of antibodies could help to create a universal vaccine for influenza.
Broadly neutralizing antibodies are small proteins produced by the immune system to bind to many strains of viruses and other invaders such as bacteria and fungi, and kill them. In influenza, they bind to the "stalk" or "stem" of the virus, which is similar in all influenza virus strains, rather than the "head," which is the target of most antibodies.
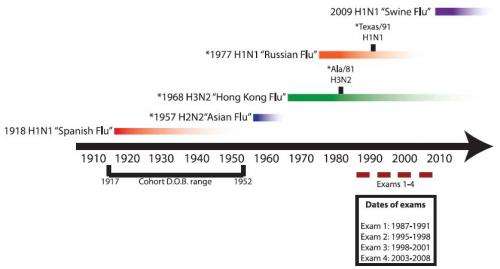
In the new study, Matthew Miller of the Icahn School of Medicine at Mount Sinai, New York, and a team of microbiologists, obtained blood serum samples taken from 40 individuals over a period of two decades from 1987 to 2008, and looked at the immune response of the broadly neutralizing antibodies in samples obtained from the subjects. The subjects were born in the years 1917 to 1952 and were part of the Framingham Heart Study.
Pandemic strains of virus have very different heads to non-pandemic strains of virus, and the researchers thought that when the body is exposed to these novel viruses it might produce more broadly neutralizing antibodies to attack the stalk of the virus. To test this hypothesis, the study included subjects who had been exposed to the Asian flu pandemic (H2N2) in 1957, Hong Kong flu (H3N2) in 1968, and/or to H1N1 in the Russian flu pandemic of 1977.
Their results showed that levels of the broadly neutralizing antibodies rose over time in all the subjects, but those who had been exposed to the H2N2 or H1N1 influenza viruses had significantly higher levels than the other participants, and those who had been exposed to more than one pandemic had even greater levels of the antibodies. They also found that in subjects exposed to pandemics, their levels of antibodies targeting the virus head increased over time, even though they had only been exposed to the pandemic virus once.
The results suggest that vaccines mimicking the body's reactions to many types of virus, especially those with similar stalks but different heads, could provide much greater protection. Dr Miller said that if we could create a vaccine that mimics the response to a pandemic "we may be able to generate a universal flu vaccine."
The paper was published on August 14th in Science Translational Medicine.
More information: Neutralizing Antibodies Against Previously Encountered Influenza Virus Strains Increase over Time: A Longitudinal Analysis, Sci Transl Med 14 August 2013: Vol. 5, Issue 198, p. 198ra107 DOI: 10.1126/scitranslmed.3006637
Abstract
Antigenic diversity shapes immunity in distinct and unexpected ways. This is particularly true of the humoral response generated against influenza A viruses. Although it is known that immunological memory developed against previously encountered influenza A virus strains affects the outcome of subsequent infections, exactly how sequential exposures to antigenically variant viruses shape the humoral immune response in humans remains poorly understood. To address this important question, we performed a longitudinal analysis of antibody titers against various pandemic and seasonal strains of influenza virus spanning a 20-year period (1987 to 2008) with samples from 40 individuals (birth dates, 1917 to 1952) obtained from the Framingham Heart Study. Longitudinal increases in neutralizing antibody titers were observed against previously encountered pandemic H2N2, H3N2, and H1N1 influenza A virus strains. Antibody titers against seasonal strains encountered later in life also increased longitudinally at a rate similar to that against their pandemic predecessors. Titers of cross-reactive antibodies specific to the hemagglutinin stalk domain were also investigated because they are influenced by exposure to antigenically diverse influenza A viruses. These titers rose modestly over time, even in the absence of major antigenic shifts. No sustained increase in neutralizing antibody titers against an antigenically more stable virus (human cytomegalovirus) was observed. The results herein describe a role for antigenic variation in shaping the humoral immune compartment and provide a rational basis for the hierarchical nature of antibody titers against influenza A viruses in humans.
© 2013 Medical Xpress














