Researchers use new genetic mapping technique to identify two genes that can cause vision loss in seniors
Researchers at the University of Michigan pinpointed two interacting genes that, when defective, can greatly increase the risk of macular degeneration, the leading cause of vision problems in the elderly.
Goncalo Abecasis, a University of Michigan professor and the lead author of the report, said much of the discovery's success is from a new method of gene mapping that uses ancestry-matched controls from existing gene databases – something that had never been done before.
"We showed this new approach could work," said Abecasis from the U-M School of Public Health. The findings will be posted online Sept. 15 in the journal Nature Genetics.
The new approach searched already sequenced individuals with similar ancestry to macular degeneration cases U-M researchers were studying. Matches were then placed in the control group, increasing the ability to pinpoint genetic variants and ultimately leading to the discovery.
Abecasis said he hopes researchers will take the new finding he and his colleagues gleaned and use it to develop new treatments or a cure for macular degeneration. The statistical methods for matching ancestry used in this study could be duplicated in genetic studies of other diseases, potentially opening up new realms of discovery.
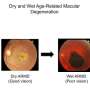
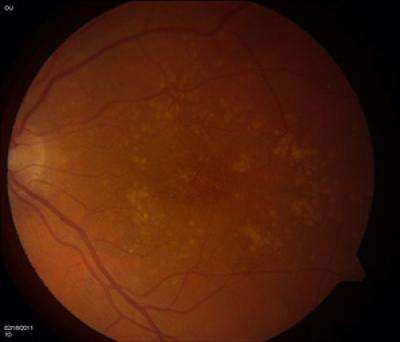
Age-related macular degeneration is a leading cause of vision loss for seniors. It is caused by the deterioration of a small, central part of the retina. It affects the ability to read, drive and carry on day-to-day activities. Currently there is no cure.
The University of Michigan has long been conducting genetic research into the causes of macular degeneration and has a large database of patient volunteers recruited at the Kellogg Eye Center.
This study involved 2,335 cases and 789 controls, with most samples originating from the Kellogg Eye Center. The research was supported in part by grants from the National Institutes of Health (National Eye Institute, National Human Genome Research Institute).
More information: Identification of a rare coding variant in complement 3 associated with age-related macular degeneration, DOI: 10.1038/ng.2758