Racing sperm to boost results of in vitro fertilization

The World Health Organization estimates more than 70 million couples worldwide are unable to conceive each year, with close to a third of those cases attributable solely to issues with male fertility—including low sperm count and low sperm motility (a limited ability to swim). Now, with recently published data showing encouraging results, researchers at Worcester Polytechnic Institute (WPI) have received a grant from the National Science Foundation (NSF) to refine a new device that races sperm through a microscopic obstacle course to select those most likely to successfully fertilize an egg.
In vitro fertilization (IVF) is the most widely used assisted reproductive technology and can help overcome problems with male fertility. Its potential for success is significantly enhanced when sperm can be sorted so only the healthiest calls and the best swimmers are used. Unfortunately, current clinical techniques for sorting sperm are inefficient or are likely to damage sperm DNA.
To solve this sorting problem, a new approach has been developed that uses advanced mathematical models and high-powered computer simulations to analyze and predict how sperm swim under varying conditions. The approach is the product of a novel research collaboration between Erkan Tüzel, PhD, assistant professor of physics at WPI, and Utkan Demirci, PhD, assistant professor of medicine and health sciences and technology at Divisions of Biomedical Engineering and Infectious Diseases, Brigham and Women's Hospital, Harvard Medical School.
Demirci's lab is developing microfluidics (small devices sometimes referred to as "lab on a chip") to test new ways to sort sperm. Tüzel and his team use the physics of fluid dynamics and customized algorithms to model how sperm cells move through narrow channels. Funded by a new three-year, $293,000 grant from the NSF, Tüzel will use simulations to optimize the design of the sperm-sorting chip. As the simulations and computer modeling progress, Demirci's lab will build and test new microfluidic chips based on Tüzel's refinements.
"We are grateful to the National Science Foundation for this grant," Tüzel said. "With our collaborators, we hope to be able to build on our research to develop clever microfluidic designs that will be even more effective in sorting sperm and improving the success of assisted reproductive technologies."
Demerici added, "the NSF has supported a true interdisciplinary effort at the convergence of medicine and fluid dynamics addressing a significant reproductive medicine challenge by a new type of thinking for sorting cells."
There are now two standard techniques used to separate out the most motile sperm. One is called the swim-up technique, in which sperm swim from a droplet of liquid into a denser layer of media; the cells reaching the denser layer most quickly are recovered and used for IVF. In the other technique, known as density gradient separation, a sperm sample is placed in a tube filled with layers of liquid of different densities. When the tube is spun in a centrifuge, the strongest swimming sperm are able to cross into the denser liquids.
Neither of these methods is particularly effective at dealing with low sperm counts or low sperm motility; in addition, the density gradient technique is known to cause damage to sperm DNA. Also, both techniques use chemical agents that may damage cells. So Tüzel and Demirci have taken a different approach.
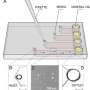
The microfluidic device they are collaborating on uses no chemicals or centrifugation. It relies only on the cells' own ability to move through channels. Within the female reproductive tract, sperm swim through a variable fluidic environment with watery micro-channels that help guide them to the egg. In the new device, sperm are placed at one end of a narrow channel and allowed to swim toward the other end. After a set period of time (called the incubation period), the sperm that have made it to the finish line are removed from the channel's end. The idea is to mimic what happens naturally.
In May, Tüzel and Demirci reported results of initial studies in the paper "Exhaustion of Racing Sperm in Nature Mimicking Microfluidic Channels During Sorting" published in the journal Small. This work combined mathematical modeling and the actual results of human and mouse sperm cells tested in a prototype of the microfluidic chip. The team found that the microfluidic channels yielded sperm with significantly higher motility and produced samples with a greater concentration of highly motile sperm than either the swim-up or density gradient techniques. This study showed the potential for developing a new technique for sorting sperm that could effectively, reliably, and safely separate out the most active swimmers. The coarse-grained model Tüzel's group built accurately recapitulated experimental results, and made predictions on the exhaustion time.
"To obtain quantitative agreement with experiments, our simulations predicted an exhaustion time of about 30 minutes for mouse sperm, whereas for human sperm the exhaustion time is more than an hour," Tüzel said. "To the best of our knowledge, this is the first attempt to quantitatively estimate exhaustion time of sperm using modeling coupled to microfluidics experiments."
Next, Tüzel's group will refine the model of sperm locomotion to produce a more realistic simulation, and model differing designs of the microfluidic channels to improve the yield of healthy and motile sperm. "We know, for example, that through hydrodynamic interactions, sperm synchronize their movements to swim more efficiently," Tüzel said. "And we are starting to understand more about how the presence of walls can impact the speed at which multiple sperm move. But we are in need of novel, efficient approaches to model complex interactions between groups of swimming sperm, and how they interact with their surroundings. That is our focus through the new grant."