Building blocks help silence genes
Polycomb-group (PcG) proteins play an important role in controlling gene expression. Complexes containing PcG proteins are thought to inhibit or 'silence' gene activity by localizing to specific targets in the genome and remodeling how DNA is wound up into chromosomes, but the exact mechanism by which these complexes repress gene activity remains poorly understood. Kyoichi Isono, Haruhiko Koseki and colleagues from the RIKEN Center for Integrative Medical Sciences have now pinpointed the part of a critical PcG protein complex that is essential for maintaining a robust yet reversible gene repression program during both mammalian development and cancer progression.
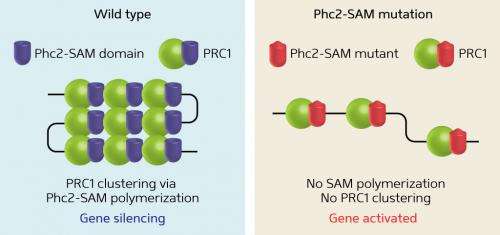
Isono, Koseki and their colleagues set out to identify the formation mechanism of a cluster of PcG proteins known as Polycomb-group repressive complex-1 (PRC1). They focused their attention on a particular domain within one of the molecules in the PRC1 complex: the sterile alpha motif (SAM) of polyhomeotic-like protein 2 (Phc2). The SAM domain helps to keep Phc2 proteins in the same orientation, facilitating head-to-tail, building-block-like linking of repeated copies of the PRC1 complex (Fig. 1).
The team created human cells designed to express Phc2 carrying a mutation in the SAM domain. This mutation prevented PRC1 binding but did not affect the basic assembly of each complex. Nonetheless, the researchers observed a substantial reduction in PRC1 cluster or 'body' formation in the cell nucleus, indicating that the construction of linked repeats of PRC1, driven by SAM domain polymerization, may be necessary for proper functioning of the complex. In embryonic mice genetically engineered to possess a mutant SAM domain, Isono, Koseki and their team observed defects in the developing skeleton, further supporting the notion that the aberrant PRC1 bodies were not maintaining proper gene regulation in the absence of the SAM domain.
The findings illuminate a key aspect of mammalian embryo formation. According to Isono, however, the results could also have implications that go well beyond the understanding of basic developmental processes. "The repressive function of PRC1 has a strong impact on not only pluripotency and differentiation of stem cells but also tumorigenesis," he says. "The mechanism that underlies SAM polymerization and depolymerization could be useful for regenerative medicine and cancer therapy."
A particularly exciting potential therapeutic target might be compounds that destroy the interactions between SAM domains, which could halt tumor growth in cancer patients. "I believe we can develop such drugs by conducting microscopic screenings for PcG body morphology," Isono says.
More information: Isono, K., Endo, T. A., Ku, M., Yamada, D., Suzuki, R., Sharif, J., Ishikura, T., Toyoda, T., Bernstein, B. E. & Koseki, H. "SAM domain polymerization links subnuclear clustering of PRC1 to gene silencing." Developmental Cell 26, 565–577 (2013). dx.doi.org/10.1016/j.devcel.2013.08.016