Gut reaction: Scientists study factors influencing intestinal microbes
Study results from Texas A&M University and University of North Carolina School of Medicine scientists on the effect of diet complexity and estrogen hormone receptors on intestinal microbiota has been published in the September issue of Applied and Environmental Microbiology.
To date, research has shown that promoting the growth of certain beneficial intestinal microorganisms can help to improve overall health. The study was to determine the effect of certain factors on intestinal microbiota.
"In this study, we wanted to determine if steroid hormone nuclear receptors, specifically estrogen receptor beta, affect the composition of intestinal bacteria," said Dr. Joseph Sturino, lead researcher in the nutrition and food science department at Texas A&M's College of Agriculture and Life Sciences, College Station.
"Some steroid hormones, like estradiol, and dietary phytoestrogens are known to influence the development of chronic gastrointestinal inflammation and estrogen-responsive cancers of the breast, prostate and colon," Sturino said.
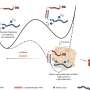
Some of these effects are the result of differential and tissue-specific gene regulation by estrogen receptor beta, Sturino said. That aspect of the study was the focus of the lab work performed by Dr. Clinton Allred, also in the college's nutrition and food science department and a collaborator on the published study.
They hypothesized that some estrogenic regulatory signals are mediated, in part, by the activity of microorganisms present in the gut and that diet modification can be used to change those.
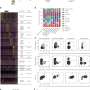
In order to investigate the effects of both receptors and diet on intestinal microorganisms, the scientists initially raised female mice on a fiber-rich diet containing plant-derived estrogenic compounds called isoflavones, comprising a complex diet. The animals were then fed an isoflavone-free diet that was rich in highly refined sugars for two weeks, comprising a simple diet. The composition of the fecal bacteria was surveyed over the course of the study.
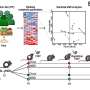
"As you might expect, significant differences were found between the fecal microorganisms of mice fed a biochemically complex diet containing isoflavones and those that were fed a simple diet that lacked isoflavones," he said. "Interestingly, however, we also found that the microorganisms differed between mice that expressed estrogen receptor beta and those that did not."
Distinct patterns for Lactobacillales were exclusive to and highly abundant among mice fed a complex diet containing isoflavones, Sturino explained.
"Some Lactobacillales have probiotic function when taken in adequate numbers in food or dietary supplements, so indigenous species might also act to promote gut health," he said.
In contrast, he noted, the relative diversity of Proteobacteria increased significantly following the transition to the simple, isoflavone-free diet. Proteobacteria includes a number of species commonly associated with intestinal disease, including Escherichia, the "E" in E. coli O157:H7, and salmonella.
These and other study results demonstrated that steroid receptor status and diet complexity might play important roles in microbiota maintenance, Sturino said.
"While the balance and content of microorganisms in the gut changes as we age, we are only now learning how our genetics and dietary choices affect our health by modifying the composition and activity of these microorganisms," he said.
In the long term, Sturino believes that this study will aid in the development of novel probiotics, prebiotics, nutritional strategies and pharmaceuticals to improve overall health by promoting the growth and activity of beneficial intestinal microorganisms.