Photopharmacology: Optoswitches turn pain off and sight on
Photoreactive compounds developed by scientists of Ludwig-Maximilians-Universitaet (LMU) in Munich directly modulate nerve-cell function, and open new routes to the treatment of neurological diseases, including chronic pain and certain types of visual impairment.
All modes of sensory perception are based on communication between nerve cells. Both the response to the primary stimulus and the transmission of the resulting signal depend on the function of specialized receptor proteins that are associated with the surface membranes of neurons. Many sensory neurons respond only to a single sensory modality, such as mechanical stimulation or temperature. However, Dirk Trauner, Professor of Chemical Biology and Genetics at LMU, has helped develop a method which, in principle, enables all types of neuroreceptors to be controlled by light. "This is achieved by using synthetic molecular compounds that react specifically to light as switches for natural receptors," he says. "The combination results in hybrid photoreceptors, which effectively make the nerve cells that bear them responsive to light. In the long term, we hope to use this approach as the basis for new therapies for neurological disease." Two new studies, on pain receptors and a mouse model for congenital blindness, now report significant advances toward the realization of this goal.
Synthetic opioid as a painkiller
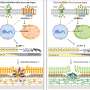
So-called opioids, natural substances related to morphine, have been used as painkillers for thousands of years. The body also synthesizes endogenous opioids, known as endorphins, which bind to the same receptors and have the same analgesic effects as morphine. By chemically modifying the synthetic opioid fentanyl, which is used clinically both as an anesthetic and as an extremely potent painkiller, Trauner and his team have succeeded in conferring photosensitivity on a particular class of opioid receptor. The modified fentanyl retains the ability to bind to Mu-type opioid receptors, but its physiological effect can be precisely controlled by light: Specifically, receptor function can be activated and deactivated by irradiating cells exposed to the compound with light of different colors, as Trauner and his colleague Matthias Schönberger report in the journal Angewandte Chemie.
The secret of Trauner's light-sensitive switches lies in their chemical structure. The agents synthesized in his laboratory all contain a characteristic chemical double bond at a specific position, which enables their shape to be altered by light. In effect, these molecules can be flipped from a bent to an extended form, and back again, depending on the wavelength of light used. Trauner explains the advantages of the method as follows: "The properties of light can be very precisely manipulated, so that we can modulate the state of the cells in a defined fashion. Furthermore, the reaction is fully reversible."
"The ability to control the functional states of opioid receptors is of particular interest because they belong to the large family of so-called G-protein-coupled transmembrane receptors (GPCRs), which make up a large fraction of the proteins targeted by pharmaceutical agents," says Matthias Schönberger, first author of the new study. "The capacity to regulate an opioid receptor by means of light will make it possible to obtain new insights into the mode of action of this eminently important class of receptors, and could offer a route to novel treatments of chronic pain syndromes," he adds.
Bypassing damaged retinal cells
The light-sensitive photoreceptors made by the rod and cone cells in the retina also belong to the GPCR class. Moreover, they are the only GPCRs that are intrinsically sensitive to light. Functional damage to these photoreceptors, or pathological loss of the cells that bear them, results in inability to register light impinging on the retina – and is responsible for various types of visual impairment and certain forms of congenital blindness. In collaboration with researchers at the University of California in Berkeley, Trauner and his group have, for the first time, succeeded in restoring visual function to mice in which retinal photoreceptive cells had degenerated. The trick was to use a new synthetic switch to confer light sensitivity on the retinal ganglion cells in these mice, which normally respond to signals from the rods and cones upstream of them.
"We demonstrated some time ago that a molecule called AAQ is capable of causing nerve cells to become light-sensitive, because it modifies the behavior of ion channels in their surface membranes," Trauner says. In the meantime, he and his group had synthesized an improved variant of the AAQ switch, which they call DENAQ. The scientists in Berkeley have now shown that DENAQ can indeed restore light responsiveness to a retina that has lost its primary photoreceptive cells, as they report in the latest issue of the journal Neuron. "From the clinical point of view, DENAQ is a great advance on AAQ, because it acts specifically on a particular type of ion channel that is essential for the transmission of signals received from the rods and cones," says Trauner. In addition, and unlike AAQ, it reacts to wavelengths present in normal daylight, to which the retina is normally sensitive. "In the future, this approach could help to restore sight in people suffering from certain forms of blindness."
"In principle, one can imagine a whole range of possible applications for photopharmacology," says Trauner. "Some GPCRs, for instance, are targeted by neurotransmitters, and their functions are compromised in diseases of the central nervous system, so it is possible that optical switches could also be of interest in the treatment of conditions such as depression or epilepsy. And beyond that, one could make use of the unparalleled spatial and temporal precision of light signaling to activate compounds such as analgesics, cytostatic drugs or anti-diabetic agents only at sites where they are required, thus minimizing unwanted side effects.
More information:
Angew Chem Int Ed Engl. 2014 Feb 12. DOI: 10.1002/anie.201309633. [Epub ahead of print]
Neuron, Volume 81, Issue 4, 800-813, 19 February 2014 DOI: 10.1016/j.neuron.2014.01.003