March 31, 2014 feature
Connecting the dots: Water diffusion MRI reveals plasticity networks in remote nonstimulated brain regions
(Medical Xpress)—Neuroplasticity (also referred to as brain plasticity, or simply plasticity) is the ability of the brain's neurons and neural networks – previously counter to what scientists believed – to change their connections and behavior when presented with a range of conditions, including neurodevelopment, novel information, sensory stimulation, and trauma. Moreover, modular brain areas can self-reorganize and perform activities differing from their typical functions. In the lab, researchers can induce site-specific plasticity through an established experimental method known as repetitive transcranial magnetic stimulation (rTMS), in which a pulsed noninvasive magnetic field roughly the same strength as a magnetic resonance imaging (MRI) scan depolarizes or hyperpolarizes the neurons in the target brain site. Compared with non-repetitive transcranial magnetic stimulation, rTMS produces longer-lasting effects that persist past after the stimulation has stopped, and has been used as a treatment modality in stroke, Parkinson's disease, depression, and other neurological and psychiatric disorders.
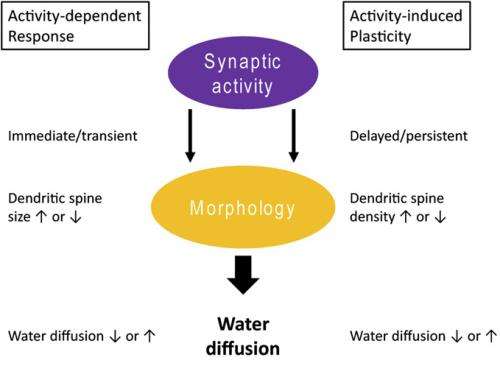
While plasticity-related changes in remote non-stimulated brain areas has been suggested by recent research, the actual physical processes causing these remote effects has remained unknown – but recently, scientists in the Human Brain Research Center at Kyoto University Graduate School of Medicine used diffusion-weighted MRI (DWI) to estimate morphological changes in cortical tissue undergoing rTMS-induced plasticity in human brains. (DWI allows researchers to investigate water diffusion, a thermal physical phenomena characterized by random motion of water molecules.) The researchers found that coupling of water diffusion between regions revealed synchronized fluctuations in the microstructural morphology connecting stimulated and remote regions, forming what they have termed multiregional water diffusion networks – networks revealed by water diffusion found in this study for the first time as indicated by the neuroscience literature – that may finally explain rTMS-induced remote plasticity. In addition, they state that DWI techniques could provide a method for evaluating the effects of brain stimulation treatments brain disorders.
Prof. Mitsunari Abe discussed the research that he, Prof. Hidenao Fukuyama and Prof. Tatsuya Mima published in Proceedings of the National Academy of Sciences. "Plasticity at the stimulated cortex and morphological change in multiple regions, including the stimulated cortex, are completely different," Abe tells Medical Xpress. Specifically, he explains, plasticity is an electrophysiological estimate of signal transduction between synapses measured to evaluate the direct rTMS effect in the stimulated cortex while morphological change is accompanied with induction of plasticity but a different estimate of additional rTMS effects.
"We know from brain slice models that repetitive brain stimulation protocols affect the stimulated site, but the effect of stimulation on non-stimulated remote regions has not been investigated and remains poorly understood," Abe continues. "When we apply the stimulation protocols to human brains in experimental and clinical settings, we need to understand the effect of stimulation on remote regions. Therefore, in this study we faced the challenge of visualizing remote effects in whole brains." Abe points out that morphological change in neural tissue microstructures is accompanied by plasticity induction, so detecting morphological change provides direct evidence that rTMS sculpts remote regions of the brain in a way similar to plasticity observed at the stimulated neurons in long-term potentiation (LTP) or long-term depression (LTD). "That's why we decided to evaluate morphological dynamics with DWI techniques."
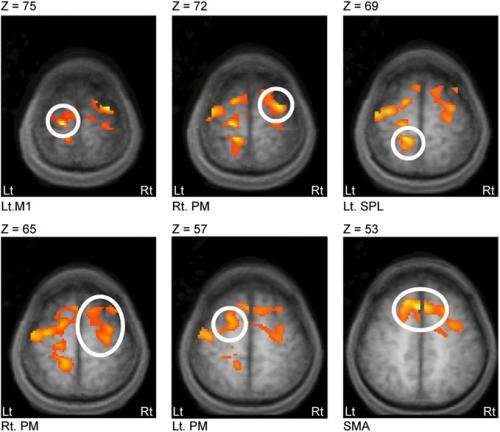
The scientists chose to investigate the left primary motor cortex (M1) using DWI techniques because, Abe says, of "a simple, practical reason: When we stimulate M1, we can measure an output from muscles - motor-evoked potentials (MEPs) to estimate plasticity in the M1 cortex." When beginning their study and based on previous reports, he notes, they measured MEPs to confirm that rTMS applied to M1 induced plasticity. "Then we tested changes in DWI before and after rTMS – the primary purpose of our study." Abe adds that the application of rTMS to M1 can be used as a therapeutic tool for cerebrovascular stroke patients with motor disabilities. "In clinical settings, we can easily apply our experimental design to evaluating therapeutic effects."
The researchers addressed these challenges in several ways. "We aimed to observe water diffusion fluctuations over time," Abe says – and since it usually takes a long time (six to nine seconds) to scan whole brains, they scanned a partial brain covering M1 and other motor-related regions. Furthermore, they measured water diffusivity along one single direction, although DWI typically measures diffusivity with at least three directions because water molecules move randomly in a 3D space. "We can assume that measuring one direction be OK," he explains, "because diffusivity should be the same in all directions in the cortex due to Brownian motion." These scanning protocols reduced the time to complete one scan and allowed repetitions of scanning brains as many times as possible, allowing the researchers to obtain the time-series data they needed to analyze network connectivity.
The study's key finding reveals how synchronized fluctuations in the morphology of microstructures between stimulated and remote regions might form hitherto unknown water diffusion networks that allow retention of plasticity-related morphological changes in remote regions. "To our knowledge, our results are the first demonstration that stronger coupling with the stimulated cortex at rest can predict a longer-lasting retention of the post rTMS effects in remote regions," Abe points out. "Moreover, our results could provide the new insight that resting networks revealed by water diffusion – consisting of the stimulated cortex and each coupled remote region – might be a neural basis that allows longer-lasting retention of the rTMS effects. Given that morphological change in neural tissue modulates water diffusion, it is therefore interpretable that synchronized fluctuations in the morphology of microstructures between regions might form networks to retain the additional effects of rTMS."
In addition, Abe continues, recent reports indicate that fluctuations in morphology occur as a second-order effect of net synaptic activity oscillations "Based on our results, we speculate that water diffusion behaviors reflect an aspect of synaptic activity that differs from vascular responses." The scientists want to understand the underlying biological systems using animal model, and have in fact started to collaborate with experts engaged in relevant animal studies.
Regarding their suggestion that DWI techniques could provide a tool to evaluate treatment effects of brain stimulation protocols in patients with brain disorders, Abe says that "so far, rTMS has been established as a therapeutic tool by modulating abnormal brain functions and recovering functions to normal conditions. However, it's still unknown whether the direct effect of rTMS to the stimulated cortex would improve brain function or additional effects of rTMS in remote regions would improve in each specific disorder." He adds that when they find target regions where greater rTMS-related changes were correlated with higher therapeutic effects, this information could provide new designs for more efficient treatment strategies to affect the target regions.
"There are at least three scenarios," Abe continues. "If the target regions are the stimulated cortex, we don't need change the stimulation site; if the target regions are remote regions and the cortices near the skull surface, we will stimulate these target cortices directly; if the target regions are remote regions and deep structures, we can't stimulate these subcortical regions directly, so we will determine the stimulation site so that the rTMS effect would be the strongest in the target regions. In the third case, when we have information about water diffusion networks we can predict before we apply rTMS protocols in patients which regions retain the post-rTMS effects, and whether non-stimulated target regions can retain the rTMS therapeutic effects. Although the biological significance of the identified networks is still unknown," he acknowledges, "information based on DWI data would be helpful for new designs."
Asked if their research might relate synchronized morphological fluctuations to neurotransmitter anomalies found in brain disorders such as unipolar or bipolar depression, Abe says that recent evidence indicates that neuromodulators such as dopamine can alter networks that synchronize neuronal firing between nodal regions. "In fact," he notes, "we plan to examine the effect of neuromodulators on water diffusion networks in healthy subjects and patients. We'll be able to answer this question when we have that data."
Moving forward, Abe tells Medical Xpress, the scientists have several planned steps planned:
- Electrophysiological estimates in animal models of additional rTMS effects in remote regions
- Do neuromodulators alter water diffusion behaviors?
- Application of DWI techniques in patients
"Plasticity is an experimental model for formation of memory," Abe concludes. "Our techniques may be helpful to visualize brain regions responsible for memory formation."
More information: Water diffusion reveals networks that modulate multiregional morphological plasticity after repetitive brain stimulation, Proceedings of the National Academy of Sciences, Published online before print on March 11, 2014, doi:10.1073/pnas.1320223111
© 2014 Phys.org. All rights reserved.