Where have all the mitochondria gone? Researchers shed light on a crucial step in fertilization
It's common knowledge that all organisms inherit their mitochondria – the cell's "power plants" – from their mothers. But what happens to all the father's mitochondria? Surprisingly, how – and why – paternal mitochondria are prevented from getting passed on to their offspring after fertilization is still shrouded in mystery; the only thing that's certain is that there must be a compelling reason, seeing as this phenomenon has been conserved throughout evolution.
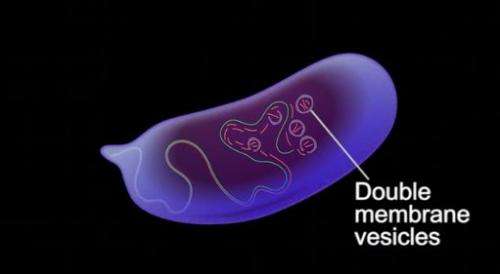
Now, Dr. Eli Arama and a team in the Weizmann Institute's Molecular Genetics Department have discovered special cellular vesicles that originate in the female fruit flies' egg and which actively seek out and destroy the father's mitochondria upon fertilization.
This study, recently published in Development Cell, may help shed light on the prevailing theories. One holds that it is an active process in which paternal mitochondria are selectively degraded by a "self-eating" system known as autophagy, in which vesicles called autophagosomes engulf the cell's unwanted structures. But the autophagy study was conducted on worms (C. elegans) whose sperm are quite different from the long, flagellated "head" and "tail" structures of both mammalian and fruit-fly sperm. The tail comprises the mitochondria: a long tube attached to, or coiled around, the tail's skeletal structure. How would the tiny autophagosome engulf such a large structure – about 2 mm long in the fruit fly?
A second theory, based mainly on mouse models, states that the absence of paternal mitochondria is due to a passive process of dilution in the sea of maternal mitochondria. But that could not explain why certain genetic markers related to autophagy were still detected on the paternal mitochondria after fertilization.
Enter the egg's special cellular vesicles. The Weizmann team, led by Ph.D. students Liron Gal and Yoav Politi in Arama's group, together with former senior intern Yossi Kalifa and former Ph.D. student Liat Ravid, and with the assistance of Prof. Zvulun Elazar of the Biological Chemistry Department, found that as soon as the sperm enters the egg, the cellular vesicles – already present in the fruit fly egg – immediately attract to the sperm like a magnet. They then proceed to disintegrate the sperm's outer membrane and separate the mitochondria from the tail section, which is subsequently cut into smaller pieces that are then "devoured" by conventional selective autophagy.
But what were these vesicles? Close observation revealed they did not resemble an autophagosome, but rather a different type of vesicle that is usually involved in a distinct pathway. Yet these vesicles carried autophagy markers. Arama: "We were not witnessing classic autophagy machinery; these structures were too large and morphologically distinct to be typical autophagosomes."
The team's findings suggest that the egg's special cellular vesicles represent a new type of system that is a unique combination of three separate biological processes whose pathways may have diverged from their classic functions.
These new discoveries, which the scientists believe hold true for other organisms with flagellated sperm, including humans, may lead, among other things, toward an understanding of why only a quarter of IVF pregnancies carry to term. It may be that this invasive procedure somehow abrogates the ability of the egg to destroy the paternal mitochondria. Arama and team hope that further research will help shed new light on a variety of issues pertaining to paternal mitochondria, with an ultimate goal of understanding mitochondrial turnover and male fertility.
More information: Paper: www.cell.com/developmental-cel … /abstract/S1534-5807%2814%2900204-4

















