Supermaterial gives rejected drugs a new chance
More than 80 percent of all drug candidates in the pharma R&D suffer from poor solubility and are therefore rejected early in the drug discovery process. Now Uppsala University researchers show that the new material Upsalite, has great potential for development of new formulations of these rejected drugs.
"These findings could allow the pharma companies to open up large libraries of potent drug candidates earlier discarded due to solubility problems and try them again together with Upsalite," says Maria Strømme, Professor at the Division of Nanotechnology and Functional Materials at Uppsala University.
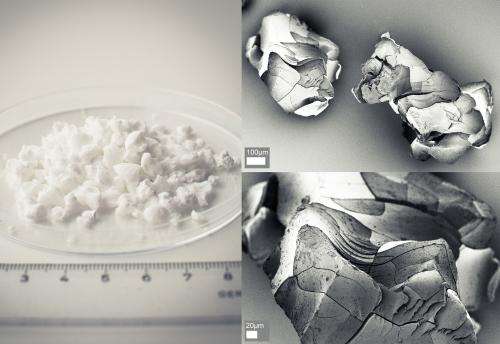
The new material was presented last year by her research group. The material had the highest surface area ever measures för an alkali earth metal carbonate; 800 square meters per gram.
In a new study, published in International Journal of Pharmaceutics the researchers show that the extremely porous magnesium carbonate Upsalite may provide a great potential for the development of new drug formulations of active pharmaceutical ingredients with poor aqueous solubility.
About 40 percent of newly marketed drugs have poor solubility and more than 80 percent of drug candidates in the R&D pipeline fail because of solubility problems. These drugs subsequently have low bioavailability (i.e., they are difficult for the body to take up), which limit their therapeutic efficacy and hence seldom leads to new pharmaceuticals.
In the new study the pain killer Ibuprofen was used as a model substance for poorly soluble drugs and it was shown that crystallisation of the drug was completely supressed in the formulation, resulting in both a higher solubility and a three times faster dissolution rate of the drug.
"We think that the narrow pores hinder the drug molecules from organizing in a poorly soluble crystalline form and are forced to maintain an amorphous structure that is easier for the body to absorb," says Johan Gómez de la Torre.
More information: Peng Zhang, Johan Forsgren, Maria Strømme. "Stabilisation of amorphous ibuprofen in Upsalite, a mesoporous magnesium carbonate, as an approach to increasing the aqueous solubility of poorly soluble drugs." International Journal of Pharmaceutics. Volume 472, Issues 1–2, 10 September 2014, Pages 185–191. www.sciencedirect.com/science/ … ii/S0378517314004451