Replacing myelin protects nerve cells, restores function in mouse model of multiple sclerosis

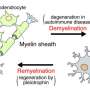
Using a mouse model of multiple sclerosis (MS), a research team led by UC San Francisco scientists has demonstrated for the first time that regenerating myelin – the fatty insulating sheath surrounding neural fibers that is destroyed in the disorder – can both protect neurons from damage and restore lost function.
"The key thing we learned from this study is that if we can design therapies that promote remyelination – especially when myelin has been damaged by inflammation as it is in MS – we can prevent neuronal loss and restore function," said UCSF's Jonah R. Chan, PhD, the senior author of the new study, which was published September 27, 2016 in the open-access journal eLife. "This is something I and other investigators have wanted to promise to MS patients, but we simply didn't have the data."
In MS, the immune system somehow goes awry and attacks myelin, compromising the efficient transmission of electrical impulses among brain cells. This leads to a range of progressively worsening symptoms, including vision problems, numbness, weakness, and difficulty walking.
There is increasing evidence that, in addition to its insulating properties, myelin also provides metabolic support to axons, the wire-like nerve-cell extensions it ensheathes. In MS, as myelin continually degrades, axons also degenerate, ultimately causing nerve cells to die off completely. It is this degeneration that is thought to be responsible for the chronic disability and progression of symptoms seen in MS.
Exploring a Key Hypothesis About Myelin
In light of this disease process, it would seem logical that restoring myelin would protect axons, which in turn would protect neurons overall and help to maintain normal brain function, and many scientists and pharmaceutical firms are pursuing MS treatments based on just that premise.
But Chan, the Debbie and Andy Rachleff Distinguished Professor of Neurology at UCSF, said he was frankly surprised to learn that there were no hard data to support this key hypothesis, and he therefore assembled an international team of scientists to conduct the new study.
The researchers used the so-called EAE (experimental autoimmune encephalomyelitis) model of MS, in which mice are injected with a protein found in myelin, which incites an inflammatory autoimmune response with physiological and behavioral consequences similar to those seen in human MS.
In previous UCSF research employing high-throughput drug-screening technology that Chan designed, he and colleagues discovered a cluster of compounds, all of which target proteins known as muscarinic receptors, that promote remyelination – the process by which cells known as oligodendrocytes will rewrap themselves around axons to repair the myelin sheath. Within this collection was an over-the-counter antihistamine called clemastine.
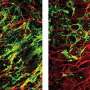
In the current study, the researchers simultaneously injected mice with both clemastine and the protein that induces EAE, and they indeed found that these mice had less severe MS-like symptoms, and that some remyelination took place in the brain and spinal cord. Axons also appeared to be protected from degeneration in mice that received clemastine.
Mechanism of Action Porly Understood
But clemastine's mechanism of action is poorly understood, Chan said, and pharmacologically speaking it is a "dirty" compound: in addition to its effects on histamine and muscarinic receptors, it interacts with many other types of receptors, and it affects many types of cells besides oligodendrocytes. So it wasn't possible for the researchers to disentangle whether the less-severe symptoms and evidence of new myelin seen in clemastine-treated mice were truly the direct result of a specific effect of the drug on oligodendrocytes, or was instead due to some broader, indirect effect, such as dampening the overall inflammatory response.
To address this question, the team conducted a series of experiments to identify which oligodendrocyte receptor clemastine might be acting on. They obtained numerous "knockout" mice, each lacking a specific receptor target, and systematically tested the drugs identified in the previous high-throughput screen in these mice. Ultimately, the team identified M1R (muscarinic acetylcholine receptor 1 subtype) as the target for clemastine and other anti-muscarinic compounds identified in the original screen, and determined that M1R was a potent inhibitor of oligodendrocyte differentiation from precursor cells, which is critical for remyelination.
No drugs exist that specifically and potently block M1 without affecting other muscarinic receptors, so the group continued using a genetic approach, employing knockout mice lacking the M1 receptor specifically in oligodendrocytes and testing these mice in the EAE model of MS.
In these mice, there was significant remyelination, axons were protected from degeneration, and function was restored, even when EAE inflammation was at its peak. Because of the precision of the gene knockout, the researchers are confident that all these effects followed from the absence of the M1 receptor in oligodendrocytes, which appears to have a profound inhibitory effect on remyelination.
The next step, Chan said, will be to try to design a "first-in-class" M1-blocking drug and to test its efficacy in animal models, and ultimately in MS patients. To that end, Chan and co-author Ari J. Green, MD, associate professor of neurology and clinical director of the UCSF Multiple Sclerosis Center, have partnered with Daniel Lorrain, PhD, head of biology, and Brian Stearns, PhD, head of chemistry, at San Diego-based Inception Sciences to advance this project.
In addition to Chan and Green, UCSF authors of the new study include former postdoctoral fellow Feng Mei, PhD, now at Third Military Medical University, in Chongquing, China; postdoctoral fellow Klaus Lehmann-Horn, MD; Yun-An A. Shen; Kelsey A. Rankin; Kara Pekarek, a former research associate, now at Armo Biosciences; Sharon A. Sagan, research associate; H. Christian Von Büdingen, MD, associate professor of neurology; Steven P.J. Fancy, DVM, PhD, assistant professor of pediatrics; and Scott S. Zamvil, MD, PhD, professor of neurology.
They were joined by researchers from the Third Military Medical University; Inception Sciences; the University of Vermont; the National Institutes of Health (NIH); and Texas Tech University Health Sciences Center.
The work was funded by the National Multiple Sclerosis Society; Target ALS; The National Institutes of Health; and the Rachleff Endowment.
"Now that we've shown we can promote repair during the peak inflammation period, and that new myelin may remain stable," Chan said, "we can now say to MS patients that focusing on this remyelination space has the potential to not only restore function, but to improve their quality of life."
More information: Feng Mei et al. Accelerated remyelination during inflammatory demyelination prevents axonal loss and improves functional recovery, eLife (2016). DOI: 10.7554/eLife.18246