Stroke study reveals key target for improving treatment and suggests that Gleevec may help
Research in mice shows why tPA carries risks as well as benefits, and suggests that Gleevec or other drugs could prevent problems
For over a decade, the drug called tPA has proven its worth as the most effective emergency treatment for the most common kind of stroke. But its promise is blemished by two facts: tPA can cause dangerous bleeding in the brain, and its brain-saving power fades fast after the third hour of a stroke.
Now, a new paper published online in Nature Medicine reveals why tPA has these limitations. It also gives tantalizing evidence about how those problems might be overcome, if a stroke victim first takes a drug currently used to treat leukemia.
The researchers, from the University of Michigan and the Ludwig Institute at the Karolinska Institutet in Stockholm, Sweden, emphasize that it's still too early to apply their findings — made in mice — to the treatment of stroke victims everywhere.
But the Karolinska Institutet team will soon begin a clinical trial to test the theory in humans, using the leukemia drug known as imatinib (Gleevec). In mice, that drug greatly reduced bleeding, even if tPA wasn't given until five hours after a stroke began.
The new paper details a series of molecular and cellular experiments conducted by the two teams, which began collaborating after hearing of each other's work.
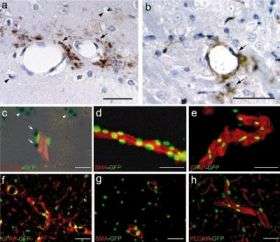
They report that tPA apparently causes its risk of bleeding, and leakage of fluid within the brain, by accident. The culprit: tPA's tendency to act upon a protein called PDGF-CC, and the PDGF-alpha receptor that it binds to. This interaction causes the usually impervious "blood-brain barrier" to become porous, leading to leakage. Gleevec inhibits the PDGF-alpha receptor, apparently counteracting tPA's effect.
This unwanted effect on the blood-brain barrier appears to be unrelated to tPA's main job, which is to break down clots that have lodged in the brain's blood vessels, cutting off blood supply to the area and starving brain tissue until it begins to die.
Such clots cause 80 percent of the 15 million strokes that occur each year worldwide. Five million people die, and 5 million more are permanently disabled, by strokes each year, according to the World Health Organization.
"Our findings may have immediate clinical relevance, and could be applied to find new treatments that will benefit stroke patients," says senior author Daniel Lawrence, Ph.D., professor of cardiovascular medicine in the U-M Medical School and member of the U-M Cardiovascular Center. "By better understanding how the brain regulates the permeability of the blood-brain barrier, and how tPA acts upon that system, we hope to reduce the risks and increase the time window for stroke treatment."
Ulf Eriksson, Ph.D., the leader of the team at the Ludwig Institute at the Karolinska Institutet, comments, "Our research group identified the growth factor PDGF-CC ten years ago and we are now very excited having unraveled a mechanism in the brain involving this factor, which potentially will be a revolution in the treatment of stroke. Together with our clinical colleagues at the Karolinska University Hospital in Stockholm we are now rapidly continuing to explore this exciting possibility in clinical trials involving stroke patients."
Eriksson and Lawrence collaborated with a number of colleagues — including lead author Enming Joe Su, Ph.D. of U-M and teams at the University of Maryland and Emory University — to perform the study.
Some of the mice in the study lacked the natural tPA that the body makes on its own. Some of the mice underwent a procedure that simulated the effects of a clot-based, or ischemic, stroke.
The researchers first demonstrated in non-stroke mice that tPA and PDGF-CC appeared to act on the same target, but that to cause the blood-brain barrier to leak, they both had to be injected directly into the brain side of the barrier. So, something else must be causing tPA to produce this effect when it is delivered through the bloodstream, which is how it is given during stroke treatment.
That's where the PDGF-alpha receptor comes in. The team looked at the activation of these receptors in the side of the brain where a stroke occurred, compared with the other side, in both normal mice and mice that were deficient in natural tPA. This experiment confirmed that the receptor is crucial to the blood-brain barrier permeability that is caused by tPA activation of PDGF-CC.
Knowing that imatinib is a drug that inhibits the PDGF-alpha receptor, the researchers then tested the drug's effect as a preventive agent.
First, they induced strokes in the mice, then gave some of the mice a dose of imatinib one hour after the stroke began. The mice that received the drug had 33 percent less leakage than those that didn't, and 72 hours later, the mice that received imatinib had 34 percent less damage to the brain than the others.
Finally, the researchers tested the effect of imatinib as a pre-treatment to be given before tPA, to protect against bleeding in the brain. They gave imatinib to mice one hour after the stroke began, and then waited until five hours had elapsed after the start of the stroke before giving tPA.
The level of bleeding in the brain was measured by assessing the amount of hemoglobin in the stroke-affected side of the brain. Mice that had received imatinib before tPA had 50 percent less hemoglobin than those that had not received pretreatment.
This last experiment is especially encouraging for its potential to be translated into clinical practice, says Lawrence. Stroke diagnosis often takes hours, especially if a patient delays getting to the hospital or needs to be transferred to a hospital that offers diagnostic brain scans and tPA treatment.
If the clinical trial in human patients in Sweden bears out the findings seen in mice, perhaps Gleevec could be given immediately upon suspicion of stroke-like symptoms, before diagnostic scans and other tests can be made to determine if a patient could benefit from tPA.
Source: University of Michigan Health System