Molecular switch found in mice could lead to future obesity treatments
A surprise discovery -- that calorie-burning brown fat can be produced experimentally from muscle precursor cells in mice -- raises the prospect of new ways to fight obesity and overweight, say scientists from Dana-Farber Cancer Institute.
Reporting in the Aug. 21 issue of the journal Nature, the researchers demonstrated that brown fat, which is known as the "good" form of fat -- so called because it burns calories and releases energy, unlike "bad" white fat that simply stores extra calories -- can be generated from unspecialized precursors that routinely spawn skeletal muscle.
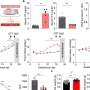
The team led by Dana-Farber's Bruce Spiegelman, PhD, showed that a previously known molecular switch, PRDM16, regulates the creation of brown fat from immature muscle cells. They also determined that the process is a two-way street: Knocking out PRDM16 in brown fat cells can convert them into muscle cells. However, Spiegelman called the latter an "experimental lab trick" for which he currently envisions no practical applications.
The "huge surprise" of the study results, he said, was that muscle precursor cells known as "satellite cells" are able to give birth to brown fat cells under the control of PRDM16.
Spiegelman said the finding confirms that PRDM16 is the "master regulator" of brown fat development. The confirmation will spur ongoing research in his laboratory, he said, to see if drugs that rev up PRDM16 in mice -- and potentially, in people -- could convert white fat into brown fat and thereby treat obesity. Another strategy, he said, might be to transplant brown fat cells into an overweight person to turn on the calorie-burning process.
"I think we now have very convincing evidence that PRDM16 can turn cells into brown fat cells, with the possibility of combating obesity," said Spiegelman, the senior author of the paper. The lead author is Patrick Seale, PhD, a postdoctoral fellow in the Spiegelman lab.
Another paper in the same issue of Nature described a different trigger of brown fat production, a molecule called BMP7. A commentary in the journal by Barbara Cannon, an internationally recognized researcher in the biology of fat cells at the University of Stockholm, said that the two reports "take us a step closer to the ultimate goal of promoting the brown fat lineage as a potential way of counteracting obesity."
The Spiegelman group has long studied fat cells both as a model for normal and abnormal cell development, which relates to cancer, and also because fat cells play such a key role in the growing epidemics of obesity and diabetes.
There is much interest in brown fat's role in regulating metabolism. Rodents and human infants have abundant brown fat that dissipates food energy as heat to protect against the cold. Though human adults have little brown fat, it apparently does have a metabolic function, including the potential to be amplified in some way to combat obesity.
In 2007, Spiegelman and colleagues reported they had inserted PRDM16 genes into white fat precursors, which they implanted under the skin of mice. The PRDM16 switch coaxed the white fat precursors to produce brown fat cells instead of white. To Spiegelman, this suggested the possibility of transplanting PRDM16-equipped white fat precursors into people who are at high risk of becoming obese, to shift their metabolism slightly into a calorie-burning mode.
The new research adds another potential source of brown fat -- the muscle cell progenitors, or myoblasts, that exist in the body to replace mature muscle cells as needed. The progenitors, which can be thought of as "adult stem cells," are committed to becoming specialized muscle cells when activated by appropriate signals, or, as the study revealed, brown fat cells when PDRM16 is turned on. The PRDM16 trigger "is very powerful at what it does," said Spiegelman, who is also a professor of cell biology at Harvard Medical School.
Source: Dana-Farber Cancer Institute