This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New model for in vitro production of human brown fat cells lays groundwork for obesity, diabetes cell therapy
Brown adipocytes are specialized cells that can use energy to produce heat. This property makes them attractive tools for the treatment of diseases like obesity and type 2 diabetes. Until recently, this therapeutic potential was constrained by limited understanding of how brown adipocyte tissue (BAT) develops from precursors.
A team led by investigators at Brigham and Women's Hospital has identified a set of cellular signaling cues that lead up to brown adipocyte formation in mice. They then used these cues to develop a protocol that efficiently produced human brown adipocytes in vitro.
The work is published in Developmental Cell.
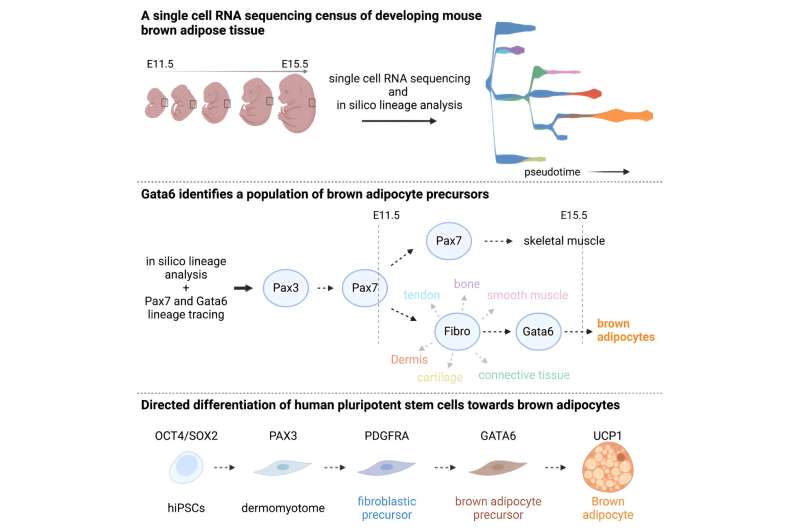
The researchers applied machine learning tools to single-cell RNA sequencing to characterize signals associated with the emergence of different types of tissue in embryonic mice. This analysis revealed a temporary stage of development that precedes BAT formation, marked by high expression of a transcription factor called GATA6. The team verified a similar pattern of expression in human BAT precursors as well.
Reproducing the sequence of signals in human pluripotent stem cells transformed them into brown adipocytes. Further, the researchers demonstrated that these cells were functional—when exposed to stimuli mimicking natural conditions in the body, they responded by increasing metabolism to produce heat. The study provides a model for reliable production of human brown fat cells in vitro.
"There is a growing body of work in preclinical models showing that grafting additional brown fat cells can improve diabetes and obesity," said corresponding author Olivier Pourquié, Ph.D., of the Brigham's Department of Pathology. "Our work could help develop cell therapy strategies for the treatment of cardiometabolic diseases."
More information: Jyoti Rao et al, Reconstructing human brown fat developmental trajectory in vitro, Developmental Cell (2023). DOI: 10.1016/j.devcel.2023.08.001