This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Understanding the cellular mechanisms of obesity-induced inflammation and metabolic dysfunction
A South Korean team has spearheaded a research endeavor that successfully uncovered the mechanisms governing inflammation and metabolic dysfunction in tissues associated with obesity. Their findings were published in Nature Communications.
According to the World Health Organization (WHO), approximately 16% of the global population is obese as of 2022. This epidemic represents one of the most rapidly escalating diseases worldwide and is serving as a leading cause of various metabolic disorders such as diabetes, hypertension, and atherosclerosis.
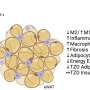
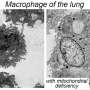
Overconsumption of nutrients prompts the infiltration of diverse types of macrophages into adipose tissue. Among them, certain macrophages play a role in clearing deceased cells and upholding tissue balance while others elicit inflammatory reactions. In patients with obesity, the population of these inflammatory macrophages escalates swiftly, exacerbating issues related to inflammation and metabolic function.
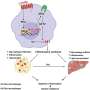
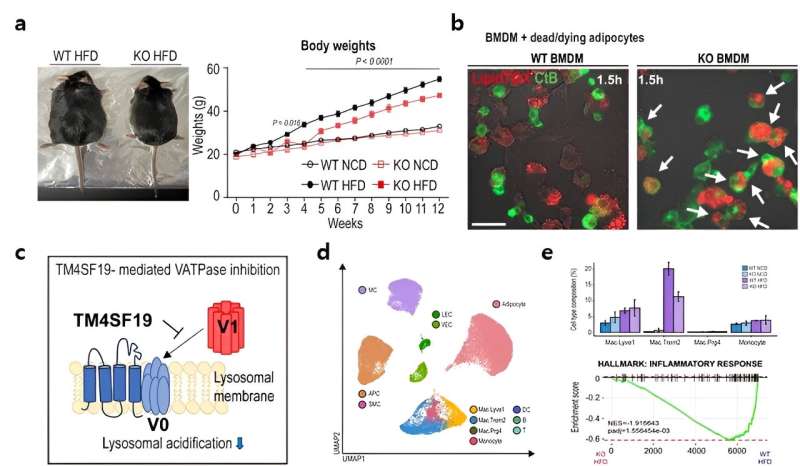
Employing animal trials, single-nucleus RNA sequencing, and intravital imaging techniques, the research team led by Professor Jong Kyoung Kim, from the Department of Life Sciences at Pohang University of Science and Technology (POSTECH), and others, scrutinized TM4SF19, a protein specifically present in inflammatory macrophages.
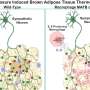
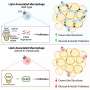
The findings revealed a notable increase in TM4SF19 levels within the adipose tissue of animal subjects subjected to a high-fat diet. Remarkably, the researchers unveiled that this protein inhibits a pump (V-ATPase) present in lysosomes, which harbor various hydrolytic enzymes and play a crucial role in lysosomal pH regulation. Consequently, this impedes the phagocytic process through which macrophages eliminate spent adipocytes.
Conversely, macrophages deficient in TM4SF19 demonstrated significantly enhanced efficacy in clearing deceased adipocytes. This not only deterred weight gain induced by a high-fat diet but also improved metabolic dysfunction by curbing tissue inflammation and insulin resistance.
The significance of this research lies in its identification of TM4SF19, present on inflammatory macrophages, as pivotal in mitigating inflammation and enhancing metabolic function in cases of obesity.
POSTECH Professor Jong Kyoung Kim stated, "We have finally unraveled the mechanism governing TM4SF19 protein's mechanism to regulate lysosomal activity. Our discoveries may open new avenues for treating obesity and related metabolic disorders."
More information: Cheoljun Choi et al, TM4SF19-mediated control of lysosomal activity in macrophages contributes to obesity-induced inflammation and metabolic dysfunction, Nature Communications (2024). DOI: 10.1038/s41467-024-47108-8