June 19, 2024 feature
This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
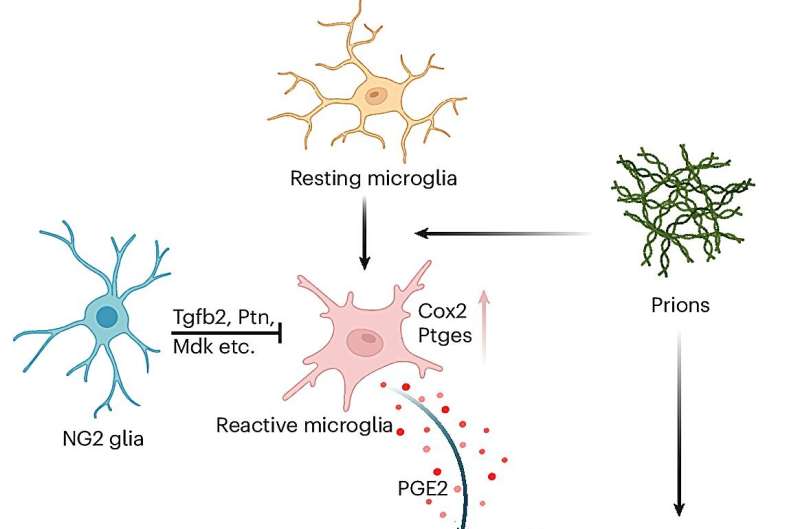
NG2 glia cells shown to protect against prion-induced neurotoxicity and neurodegeneration
Neurodegenerative diseases, including Alzheimer's disease (AD) and Parkinson's disease (PD) are highly debilitating medical conditions arising from a combination of genetic and environmental factors. Studies exploring the neural underpinnings of these diseases and how the human body typically prevents neurodegeneration could help to devise more effective treatment and prevention strategies.
Researchers at University of Zurich and Karolinska University Hospital at Solna carried out a study on mice aimed at better understanding the role of NG2 glia cells, a class of glial cells in the CNS, in prion-induced neurodegeneration. Prion diseases are a type of neurodegenerative disease in which neurotoxicity is induced by the abnormal buildup of a protein called a prion.
The researchers' findings, published in Nature Neuroscience, suggest that NG2 glia protect against the neurotoxicity and neurodegeneration linked to this build-up of prion. In addition, they provide insight into the mechanisms through which these cells could attenuate neurodegeneration.
"My interest in NG2 glia dates back to many years ago when I was investigating the regulatory mechanisms of inflammation in the central nervous system," Yingjun Liu, co-author of the paper, told Medical Xpress.
"We know that our immune system is a crucial defender of our body against infections and diseases. However, it needs to be tightly controlled because too much immune responses can be harmful to normal tissues."
Specialized cell types in peripheral organs (i.e., body parts that respond to environmental changes) are known to contribute to the rapid resolution of inflammation following the disposal of invading pathogens or damaged tissues.
Surprisingly, neuroscientists had not yet identified similar specialized cells in the CNS, despite the reported susceptibility of neurons to immune mediators released during neuroinflammation.
"My suspicion was that NG2 glia may serve as such a cell type, because they can respond to immune stimulations as quickly as classical immune cells in the central nervous system such as microglia but do not release factors that promote inflammatory responses," Liu said.
"In addition to this, earlier studies had observed possible changes of NG2 glia in the brains of patients affected by various neurological diseases. However, how they may influence disease progression was unclear."
Identifying cells that protect the CNS against neurotoxicity does not help to understand the underpinnings of specific neurodegenerative diseases. Nonetheless, it could help to devise new therapeutic interventions to cure or prevent these diseases.
"Several years ago, we developed methodologies to effectively eliminate NG2 glia from cultured brain slices and brain tissues of animal models and found that indeed NG2 glia play a crucial role in maintaining the homeostatic microglia state in the healthy brain," Liu said.
"In the current study, with the help of these methodologies, we further investigated the role of NG2 glia under pathological conditions by manipulating them in the context of prion diseases, a group of fatal brain disorders that can kill a patient within a matter of months."
The recent study by Liu and his colleagues specifically explored the role of NG2 glia in the emergence of neurodegenerative prion diseases. Prions are proteins found in nature that share the same amino acid sequences with some normal proteins that are highly abundant in the brain. This essentially means that prions and certain 'normal' proteins are encoded by the same pieces of DNA in the human genome.
"Yet prions are folded abnormally in 3D compared to their normal counterparts and have the ability to transform their normal counterparts into pathological forms," Liu explained. "In fact, many brain diseases, including the very common ones such as Alzheimer's disease, are caused by similar pathogenetic mechanisms. In our experiments, we mimicked prion diseases with two experimental systems."
In their first experimental setup, Liu and his colleagues exposed brain slices that were maintained 'alive' in an incubator to prions. Within weeks, the prions induced pathological changes resembling those that one would observe in the brains of patients from whom these cultures were extracted.
"This system enables us to test scientific hypotheses and potential treatments quickly," Liu said.
In their second experiment, the researchers exposed mice to prions, inducing a 'disease' that mimicked human prion diseases, with a similar disease trajectory. This allowed them to test their hypotheses and evaluate potential treatments in living mammals that share commonalities with humans.
"We eliminated NG2 glia from both model systems (i.e., the brain slices and mice) and compared them with the model systems with intact NG2 glia regarding the evolution of disease-relevant processes," Liu said. "We found that disease processes, especially the death of neurons after prion exposure, were accelerated when NG2 glia are absent, suggesting that these cells are protecting us against the disease."
Liu and his colleagues also carried out analyses to further validate their findings, specifically eliminating NG2 glia from both the tissues and mice. They found that the removal of these cells enhanced a biological process in microglia (immune cells in the brain), which is responsible for the production of a lipid molecule called prostaglandin E2.
"Crucially, previous studies have found that the level of prostaglandin E2 was higher in the brains of prion disease patients, although the relevance of the higher prostaglandin E2 level to the progression of the disease was unclear," Liu said. "So, we next investigated how the enhanced production of prostaglandin E2 after NG2 glia elimination in the prion disease models may be linked to the enhanced death of neurons."
Notably, the researchers observed that the lipid molecule prostaglandin E2 could bind to a protein called EP4 on the surface of neurons. This initiated a toxic signaling process that eventually killed the neurons, disrupting the normal functioning of the brain.
"Firstly, we found that NG2 glia in prion diseases are neuroprotective," Liu said. "We then also found that NG2 glia function as suppressors of prostaglandin E2 biosynthesis in microglia. Thirdly, we found that excessive production of prostaglandin E2 in the brain enhances killing of neurons by prions through binding to the EP4 receptor on neuronal surface."
Overall, the results gathered by this team of researchers highlight the protective effects of NG2 glia against prion-induced neurodegeneration, as the cells were found to inhibit the toxic effects of prostaglandin E2. In the future, their work could contribute to the development of new treatments for prion diseases and other similar forms of neurodegeneration.
"One interesting but still open question pertains how we can translate our findings from the current study into something that may eventually be helpful to patients, for example, by enhancing the beneficial effects of NG2 glia in our brain during the disease," Liu added.
"This will be relying on intensive future research on the molecular mechanisms that control the beneficial NG2 glia response during disease pathogenesis."
More information: Yingjun Liu et al, NG2 glia protect against prion neurotoxicity by inhibiting microglia-to-neuron prostaglandin E2 signaling, Nature Neuroscience (2024). DOI: 10.1038/s41593-024-01663-x
© 2024 Science X Network