Coaxing injured nerve fibers to regenerate by disabling 'brakes' in the system
Brain and spinal-cord injuries typically leave people with permanent impairment because the injured nerve fibers (axons) cannot regrow. A study from Children's Hospital Boston, published in the December 10 issue of the journal Neuron, shows that axons can regenerate vigorously in a mouse model when a gene that suppresses natural growth factors is deleted.
Adding to a previous study published in Science last year, research led by Zhigang He, PhD, of the F.M. Kirby Neurobiology Center at Children's Hospital Boston provides further evidence that axon regeneration is limited by a reduced or lost responsiveness to injury-induced growth factors -- and also suggests some ways of overcoming the problem to help people recover from brain or spinal cord injury.
In the earlier study, He and colleagues used genetic techniques to delete two inhibitors of a growth pathway known as the mTOR pathway in the retinal ganglion cells of mice. (These cells constitute the optic nerve, which carries visual input from the retina to the brain.) Removing this inhibition brought about vigorous growth in injured axons, but not in uninjured axons, suggesting that something about the injury itself helps trigger axon regeneration.
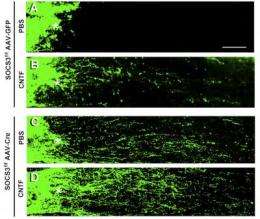
In the new study, He and colleagues used a second set of genetic techniques in mice to delete a suppressor of inflammatory signaling, known as SOCS3, in retinal ganglion cells -- and again saw robust axon growth after injury. The greatest effect was seen after one week, when there were also signs that the mTOR pathway was re-activated.
In addition, soon after injury, the team observed an increase in a growth factor called CNTF (ciliary neurotrophic factor) in the retina. When CNTF was applied directly to the eye, axons grew even more than they did with SOCS3 deletion alone. However, CNTF only modestly increased growth in mice that did not have SOCS3 deleted.
"CNTF and other cytokines [cellular signaling molecules] have been tested for promoting axon regeneration previously, but with no success," notes He. "Now we know that this is due to the tight negative control of SOCS3. Inhibiting SOCS3, using small molecule compounds or RNA interference, might allow these cytokine growth factors to be functional."
Another way of promoting axon regeneration, based on the team's findings, could be to directly stimulate the signaling pathway that SOCS3 inhibits, known as JAK/STAT, adds Fang Sun, PhD, who shares first authorship of the paper with Patrice Smith, PhD (both also of the F.M. Kirby Neurobiology Center at Children's Hospital Boston). Sun is currently testing some STAT activators.
"We are very excited by these findings," says He. "First, we are testing the combined effects of manipulating both the MTOR and JAK/STAT pathways, hoping to maximize axon growth. Second and more importantly, we are testing whether these manipulations improve functional recovery after optic nerve injury and spinal cord injury."
More information: Smith et al., SOCS3 Deletion Promotes Optic Nerve Regeneration In Vivo, Neuron (2009), doi:10.1016/ j.neuron.2009.11.021
Source: Children's Hospital Boston (news : web)














