Study: MS drugs help, but come at high cost
A new study examining the cost-effectiveness of drugs to treat multiple sclerosis (MS) in the United States finds that the health gains from these drugs come at a very high cost compared to basic therapy to control the symptoms of MS and compared to treatments for other chronic diseases. The research is published in the July 20, 2011, online issue of Neurology, the medical journal of the American Academy of Neurology.
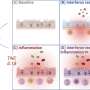
Disease-modifying drugs for MS became available in the 1990s. While they have been shown to reduce the frequency of relapses and to slow the progression of the disease, they also have significant side effects and high costs.
“Our study shows that under the current prescribing and pricing conditions in the U.S., the disease-modifying drugs account for about 50 percent of a person’s overall healthcare-related costs over 10 years and need to be brought into line so that they are not such an economic drain,” said study author Katia Noyes, PhD, MPH, with the University of Rochester in New York.
The study used data from 844 people with relapsing MS to estimate the progress of the disease over a 10-year period. In addition to examining the costs of hospital and office-based care, diagnostic tests, nursing home, home providers, health aides and lost work time, researchers also looked at whether the participants were taking any of the FDA-approved drugs for MS, including intramuscular interferon beta-1a, interferon beta-1b, glatiramer acetate and subcutaneous interferon beta-1a.
The study found that while using disease-modifying drugs for MS for 10 years resulted in modest health gains compared to basic treatment to control symptoms, the cost-effectiveness of all disease-modifying drugs was well over $800,000 per quality-adjusted life year. Quality-adjusted life year is a measure of disease burden based on the number of years of life that would be added by using a drug and the quality of life during those years. People taking intramuscular interferon beta-1a, for example, gained on average about two quality-adjusted months over 10 years compared to those who did not take disease-modifying drugs. Those taking interferon beta-1b spent an average of six out of 10 years with no relapses, compared to five years with no relapses for those who did not take disease-modifying drugs. However, the effectiveness varied significantly among individuals, with some experiencing double the improvements and being able to live independently and prolong their employment and others not observing any meaningful effect.
As context, it’s worth noting that the research did not address whether disease-modifying drugs affected the likelihood, timing or severity of MS progression, the most disabling aspect of MS; because such progression typically because noticeable many years after disease onset. The study was based on up to four years of data from observational studies, so it did not capture these outcome measures.
“Lowering the prices of these medications by 67 percent to match prices in other industrialized countries would greatly improve the cost-to-benefit ratio,” Noyes said. “The study results also support the cost-effectiveness of starting drug therapy earlier rather than waiting until a person with MS is in the later stages of the disease. Starting disease-modifying therapy earlier was associated with greater health gains and better overall cost-effectiveness. Finally, the study emphasizes the need for developing new classes of more effective medications for MS.”
After the disease-modifying drugs, the three most expensive categories related to MS were lost productivity at work, inpatient hospital admissions and in-home non-medical care.