Predicting cellular response to paclitaxel in epithelial ovarian cancer
(Medical Xpress) -- New research findings by UCD scientists published online in the Journal of Pathology may help clinicians predict how patients with high grade, serous, epithelial ovarian cancer will respond to paclitaxel chemotherapy (Taxol).
In collaboration with their colleagues in Trinity College Dublin and the Royal College of Surgeons, the UCD team highlight the central role played by a protein called mitotic arrest deficiency 2 (MAD2) in the cellular response to paclitaxel. For the first time, they have shown that low levels of this protein in epithelial ovarian cancer cells are associated with reduced progression free survival or time before relapse.
Epithelial ovarian cancer (EOC) is the leading cause of death from a gynaecological cancer in women. Each year, 334 women in Ireland are diagnosed with ovarian cancer and there are 226 deaths from the disease. With no sensitive screening test available, patients usually present with advanced stage disease. Typically, the treatment regimen consists of surgery followed by chemotherapy (paclitaxel and carboplatin) but, while 70% of patients will respond initially, many will relapse over time.
“We set about building a molecular picture of how paclitaxel interacts with epithelial ovarian cancer cells in an effort to understand why these cells become resistant over time to the chemotherapeutic drug, paclitaxel”, said Dr Fiona Furlong, Cancer Research Ireland Fellow and first author on the publication.
Paclitaxel is a microtubule stabilising agent that destroys cancer cells by interfering with the cell cycle leading to apoptosis or cell death. Specifically, it interferes with the spindle microtubule dynamics within the cell that ensures the correct alignment of chromosome pairs before they pull apart and become segregated into two daughter cells. Similar to the strings on a puppet, microtubules need to be held at the correct tension for the chromosomes to align. Anything interfering with this process will ultimately cause cell death.
“MAD2 is a key regulator of the spindle assembly checkpoint pathway and in this research we have shown that MAD2 protein expression is necessary for paclitaxel to work in ovarian cancer cells”
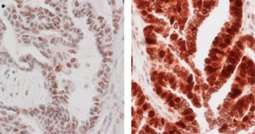
The team demonstrated that ovarian cancer cell lines expressing low levels of MAD2 protein are less sensitive to paclitaxel. They looked at the intensity of MAD2 immunohistochemical nuclear staining in 82 patient samples and found that low staining intensity levels associated with patients who relapsed soon after finishing chemotherapy.
They were also able to pinpoint for the first time that a member of a large family of gene regulators, pre-miR-433, is responsible for the down-regulation of MAD2 protein expression and consequently lessens cellular responses to paclitaxel in ovarian cancer cells.
Commenting on the significance of the findings, Dr Amanda McCann, Conway Fellow and last author on the publication said, “There are few published studies on the role of aberrant MAD2 expression and cellular drug resistance to chemotherapy in human cancer tissue samples. This work used clinical samples to identify a novel association between low MAD2 expression and reduced progression free survival in patients with high grade serous EOC. We believe that measuring MAD2 nuclear staining intensity in the clinical setting may allow clinicians to predict paclitaxel responses in women presenting with this disease.”
More information: Low MAD2 expression levels associate with reduced progression free survival in patients with high-grade serous epithelial ovarian cancer, Fiona Furlong, Patricia Fitzpatrick, Sharon O'Toole, Sine Phelan, Barbara McGrogan, Aoife Maguire, Anthony O'Grady, Michael Gallagher, Maria Prencipe, Aloysius McGoldrick, Paul McGettigan, Donal Brennan, Orla Sheils, Elaine W. Kay, John O'Leary and Amanda McCann, DOI: 10.1002/path.3035