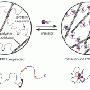
Scientists merge spider silk, human muscle to design a novel, self-assembling peptide
(Medical Xpress) -- Because of its high water content and polymer network, peptide hydrogel is a promising material for protein storage and transfer without significant loss of their biological activity. These hydrogels have potential as injectable materials for medical applications, e.g., liquid injection agents that become gelatinous in the human body to keep drugs around cancerous tumors. In this study, scientists from Kansas State University, University of Nebraska, and PNNL used two native functional sequences from spider flagelliform silk protein and a trans-membrane motif of human muscle L-type calcium channel to design a self-assembling peptide, h9e.
The h9e peptide formed two novel hydrogels in Ca2+ solution and acidic pH conditions—h9e Ca2+ hydrogel and h9e acidic hydrogel. The shear-thinning, rapid-strength-recovering h9e Ca2+ hydrogel proved to have potential for drug delivery and tissue-engineering applications and was tested on mice as an injectable adjuvant for H1N1 swine influenza virus killed vaccine. The study showed it was biologically safe, improved immune response on killed H1N1 virus antigen by approximately 70%, and induced a similar H1N1-specific IgG1 antibody response compared with an oil-based commercial adjuvant.
To assess these rationally designed peptide hydrogels, the researchers used electrospray ionization followed by analysis of the resulting ions in an LTQ-Orbitrap high-resolution mass spectrometer at EMSL. The mass spectrometry experiments were conducted to identify possible precursors of the peptide assembly and nanofiber crossing, as well as the binding mode of calcium to the peptides.
More information: Soft Matter 7(19):8905-8912. DOI: 10.1039/C1SM05157A (cover date: October 7, 2011).